Scopus, KCI, KoreaMed

Articles
- Page Path
- HOME > Surgical Metabolism and Nutrition > Volume 12(1); 2021 > Article
- Original Article Effect of Malnutrition Assessed by Comprehensive Nutritional Screening Tool on In-Hospital Mortality after Surgery for Gastrointestinal Perforation
-
Seung-Young Oh, M.D., Ph.D.1,2
 , Hannah Lee, M.D., Ph.D.1,3
, Hannah Lee, M.D., Ph.D.1,3 , Ho Geol Ryu, M.D., Ph.D.1,3
, Ho Geol Ryu, M.D., Ph.D.1,3 , Hyuk-Joon Lee, M.D., Ph.D.2
, Hyuk-Joon Lee, M.D., Ph.D.2
-
Surgical Metabolism and Nutrition 2021;12(1):1-6.
DOI: https://doi.org/10.18858/smn.2021.12.1.1
Published online: June 30, 2021
1Critical Care Center, Seoul National University Hospital, Seoul, Korea
2Department of Surgery, Seoul National University College of Medicine, Seoul, Korea
3Department of Anesthesia and Pain Medicine, Seoul National University College of Medicine, Seoul, Korea
- Corresponding author: Seung-Young Oh E-mail faun1226@gmail.com ORCID https://orcid.org/0000-0001-8281-2851
Copyright © The Korean Society of Surgical Metabolism and Nutrition
This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 2,570 Views
- 14 Download
- 2 Crossref
Abstract
-
Purpose This study examined the effects of malnutrition on in-hospital mortality after surgery for gastrointestinal (GI) perforation.
-
Materials and Methods Patients who underwent surgery for GI perforation between 2010 and 2017 were analyzed retrospectively. The preoperative nutritional status was assessed by the Seoul National University Hospital-Nutrition Screening Index, a tool that comprehensively evaluates 11 factors that reflect the nutritional status. The risk factors for in-hospital mortality after surgery for GI perforation were evaluated by univariate and multivariate analyses.
-
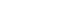
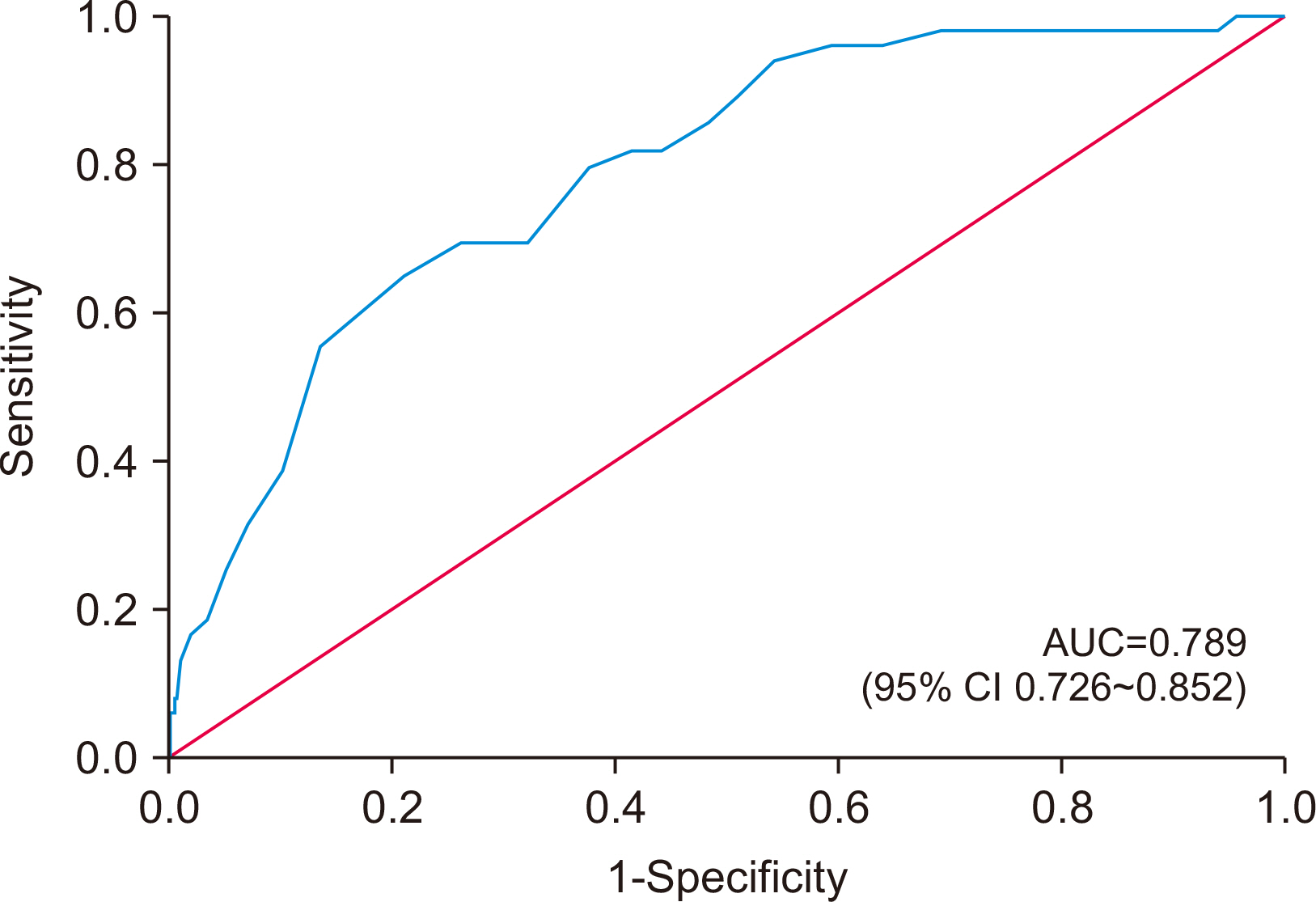
Results Four hundred and eighty-nine patients were divided into two groups: 439 patients in the survival group and 50 patients in the in-hospital mortality group. The risk of malnutrition was higher (93.6% vs. 65.9%, P<0.001) in the in-hospital mortality group than in the survival group. The preoperative albumin level was lower, and the blood urea nitrogen level was higher in the in-hospital mortality group than in the survival group. Emergency surgery, lymphoma as a cause of perforation, and fecal-contaminated ascites were also identified as factors associated with in-hospital mortality. Multivariate analyses demonstrated that a high risk of malnutrition (HR=5.71, 95% CI 1.38~26.02, P=0.017), lymphoma as a cause of perforation (HR=4.12, 95% CI 1.17~14.51, P=0.028), low preoperative albumin (HR=4.77, 95% CI 2.35~9.69, P<0.001), and high preoperative BUN (HR=1.03, 95% CI 1.01~1.05, P=0.001) had significant effects on the in-hospital mortality after surgery for GI perforation.
-
Conclusion A high risk of malnutrition assessed by the composite index was associated with in-hospital mortality after surgery for a GI perforation.
INTRODUCTION
MATERIALS AND METHODS
RESULTS
DISCUSSION
- 1. Bielecki K, Kamiński P, Klukowski M. Large bowel perforation: morbidity and mortality. Tech Coloproctol 2002;6:177-82. ArticlePubMedPDF
- 2. Pisanu A, Cois A, Uccheddu A. Surgical treatment of perforated diverticular disease: evaluation of factors predicting prognosis in the elderly. Int Surg 2004;89:35-8. PubMed
- 3. Lee JY, Lee SH, Jung MJ, Lee JG. Perioperative risk factors for in-hospital mortality after emergency gastrointestinal surgery. Medicine (Baltimore) 2016;95:e4530. ArticlePubMedPMC
- 4. Shin R, Lee SM, Sohn B, Lee DW, Song I, Chai YJ, et al. 2016;Predictors of morbidity and mortality after surgery for intestinal perforation. Ann Coloproctol 32:221-7. ArticlePubMedPMCPDF
- 5. Raguso CA, Dupertuis YM, Pichard C. The role of visceral proteins in the nutritional assessment of intensive care unit patients. Curr Opin Clin Nutr Metab Care 2003;6:211-6. ArticlePubMed
- 6. Martindale RG, McClave SA, Vanek VW, McCarthy M, Roberts P, Taylor B, et al. American College of Critical Care Medicine. A.S.P.E.N. Board of Directors. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine and American Society for Parenteral and Enteral Nutrition: executive summary. Crit Care Med 2009;37:1757-61. ArticlePubMed
- 7. Putwatana P, Reodecha P, Sirapo-ngam Y, Lertsithichai P, Sumboonnanonda K. Nutrition screening tools and the prediction of postoperative infectious and wound complications: comparison of methods in presence of risk adjustment. Nutrition 2005;21:691-7. ArticlePubMed
- 8. Kim Y, Kim WG, Lee HJ, Park MS, Lee YH, Kong SH, et al. Comparison of the impact of malnutrition by nutritional assessment and screening tools on operative morbidity after gastric cancer surgery. J Clin Nutr 2011;4:7-15. Article
- 9. Silva TA, Maia FCP, Zocrato MCA, Mauricio SF, Correia MITD, Generoso SV. Preoperative and postoperative resting energy expenditure of patients undergoing major abdominal operations. JPEN J Parenter Enteral Nutr 2021;45:152-7. ArticlePubMedPDF
- 10. Lobo DN, Gianotti L, Adiamah A, Barazzoni R, Deutz NEP, Dhatariya K, et al. Perioperative nutrition: recommendations from the ESPEN expert group. Clin Nutr 2020;39:3211-27. ArticlePubMed
- 11. Lee MS. Nutritional management in gastric cancer patients. J Korean Gastric Cancer Assoc 2003;3:172-7. ArticlePDF
- 12. Charney P. Nutrition screening vs nutrition assessment: how do they differ? Nutr Clin Pract 2008;23:366-72. ArticlePubMedPDF
- 13. Kondrup J, Rasmussen HH, Hamberg O, Stanga Z. Ad Hoc ESPEN Working Group. Nutritional risk screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr 2003;22:321-36. ArticlePubMed
- 14. Møller MB, Pedersen NT, Christensen BE. Diffuse large B-cell lymphoma: clinical implications of extranodal versus nodal presentation--a population-based study of 1575 cases. Br J Haematol 2004;124:151-9. ArticlePubMedPDF
- 15. Vaidya R, Habermann TM, Donohue JH, Ristow KM, Maurer MJ, Macon WR, et al. Bowel perforation in intestinal lymphoma: incidence and clinical features. Ann Oncol 2013;24:2439-43. ArticlePubMedPMC
- 16. Vaidya R, Witzig TE. Incidence of bowel perforation in gastrointestinal lymphomas by location and histology. Ann Oncol 2014;25:1249-50. ArticlePubMedPMC
- 17. Ahmed G, ElShafiey M, Abdelrahman H, Semary S, Elkinaai N, Romeih M, et al. Surgery in perforated pediatric intestinal lymphoma. Eur J Surg Oncol 2019;45:279-83. ArticlePubMed
- 18. Paulson S, Sheehan RG, Stone MJ, Frenkel EP. Large cell lymphomas of the stomach: improved prognosis with complete resection of all intrinsic gastrointestinal disease. J Clin Oncol 1983;1:263-9. ArticlePubMed
- 19. Baildam AD, Williams GT, Schofield PF. Abdominal lymphoma--the place for surgery. J R Soc Med 1989;82:657-60. ArticlePubMedPMCPDF
References
Figure & Data
REFERENCES
Citations

- Performance of the comprehensive nutrition screening index in predicting mortality after cardiac surgery
Jaeyeon Chung, Jinyoung Bae, Seyong Park, Dong Hyouk Kim, Youn Joung Cho, Karam Nam, Yunseok Jeon, Jae-Woo Ju
Scientific Reports.2024;[Epub] CrossRef - Efficacy of nutritional support protocol for patients with pressure ulcer: comparison of before and after the protocol
Hye Young Woo, Seung-Young Oh, Leerang Lim, Hyunjae Im, Hannah Lee, Ho Geol Ryu
Nutrition.2022; 99-100: 111638. CrossRef
Fig. 1
Seoul National University Hospital-Nutrition Screening Index (SNUH-NSI)
| R1 | R2 | R3 | |
|---|---|---|---|
| Appetite | Bad | - | Normal/good |
| Change of weight | Yes | - | No |
| Difficulty in disgesting | - | Yes | No |
| Diet type | Fluid diet | Soft blended diet of NPO | Normal regular diet |
| Serum-albumin (g/dL) | <2.8 | 2.8~3.3 | ≥3.3 |
| Serum-cholesterol (mg/dL) | - | <130 | ≥130 |
| Total lymphocyte count (cells/mm3) | <800 | 800~1,500 | ≥1,500 |
| Hemoglobin (g/dL) | - | Male <13.0 | Male ≥13.0 |
| Female <12.0 | Female ≥12.0 | ||
| C-reactive protein (mg/dL) | - | >1 | ≤1 |
| BMI (kg/m2) | <18 or ≥ 25 | 18~25 | |
| Age (years) | - | >75 | ≤75 |
| Status of malnutrition | ∙ P1; High-risk group of malnutrition; (more than 2 of R1) or (1 of R1 and more than 2 of R2) | ||
| ∙ P2; Medium-risk group of malnutrition; (1 of R1) or (more than 2 of R2) | |||
| ∙ P3; Low-risk group of malnutrition; the others |
BMI = body mass index; R = risk factor; NPO = nothing by mouth.
Patient demographics
| Survival (n=439) | In-hospital mortality (n=50) | P-value | |
|---|---|---|---|
| Age (years) | 60.4±17.0 | 65.1±14.1 | 0.057 |
| Sex (M:F) | 261:178 | 26:24 | 0.311 |
| BMI (kg/m2) | 21.8±3.5 | 21.2±3.6 | 0.228 |
| Comorbidities | |||
| Hypertension | 178 (40.6%) | 23 (46.0%) | 0.466 |
| Diabetes | 71 (16.2%) | 11 (22.0%) | 0.300 |
| Chronic liver disease | 38 (8.7%) | 9 (18.0%) | 0.035 |
| Chronic kidney disease | 38 (8.7%) | 11 (22.4%) | 0.003 |
| Coronary artery disease | 27 (6.2%) | 6 (12.0%) | 0.119 |
| Cerebrovascular disease | 18 (4.1%) | 4 (8.0%) | 0.266 |
Data are presented as mean±SD, or number (%).
BMI = body mass index.
Preoperative factors
| Survival (n=439) | In-hospital mortality (n=50) | P-value | |
|---|---|---|---|
| Nutritional risk | < 0.001 | ||
| Low risk | 30 (6.9%) | 0 (0.0%) | |
| Moderate risk | 119 (27.2%) | 3 (6.4%) | |
| High risk | 288 (65.9%) | 44 (93.6%) | < 0.001 |
| Albumin (g/dL) | 3.2±0.7 | 2.5±0.6 | < 0.001 |
| Total protein (g/dL) | 6.1±2.4 | 4.9±1.3 | 0.001 |
| Blood urea nitrogen (mg/dL) | 22.1±15.3 | 35.1±20.1 | < 0.001 |
| Creatinine (mg/dL) | 1.27±1.27 | 1.83±1.46 | 0.004 |
| C-reactive protein (mg/dL) | 13.1±12.1 | 14.9±11.1 | 0.349 |
Data are presented as mean±SD, or number (%).
Operation related factors
| Survival (n=439) | In-hospital mortality (n=50) | P-value | |
|---|---|---|---|
| Type | 0.018 | ||
| Elective | 104 (95.4%) | 5 (4.6%) | |
| Emergency | 311 (87.4%) | 45 (12.6%) | |
| Perforation site | 0.006 | ||
| Stomach | 37 (94.9%) | 2 (5.1%) | |
| Duodenum | 53 (93.0%) | 4 (7.0%) | |
| Small intestine | 99 (82.5%) | 21 (17.5%) | |
| Appendix | 43 (100.0%) | 0 (0.0%) | |
| Colon and rectum | 200 (90.9%) | 20 (9.1%) | |
| Perforation cause | |||
| Inflammatory bowel disease | 16 (3.6%) | 0 (0.0%) | 0.391 |
| Malignancy | 112 (25.8%) | 18 (13.8%) | 0.102 |
| Lymphoma | 10 (2.3%) | 6 (12.0%) | <0.001 |
| Ascites | 0.021 | ||
| Clear | 195 (92.4%) | 16 (7.6%) | |
| Turbid | 184 (90.2%) | 20 (9.8%) | |
| Fecal contaminated | 60 (81.1%) | 14 (18.9%) | 0.014 |
| Stoma formation | 178 (40.7%) | 27 (54.0%) | 0.072 |
Data are presented as number (%).
Risk factors for in-hospital mortality after surgery for gastrointestinal perforation
| P-value | HR | 95% CI | |
|---|---|---|---|
| High risk of malnutrition | 0.017 | 5.714 | 1.381~26.019 |
| Lymphoma | 0.028 | 4.115 | 1.167~14.509 |
| Hypoalbuminemia <2.8 g/dL | <0.001 | 4.769 | 2.347~9.690 |
| Blood urea nitrogen (mg/dL) | 0.001 | 1.028 | 1.011~1.045 |
BMI = body mass index; R = risk factor; NPO = nothing by mouth.
Data are presented as mean±SD, or number (%). BMI = body mass index.
Data are presented as mean±SD, or number (%).
Data are presented as number (%).

 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN Cite
Cite