Scopus, KCI, KoreaMed

Articles
- Page Path
- HOME > Ann Clin Nutr Metab > Volume 16(1); 2024 > Article
- Original Article Efficacy of high-protein diet protocol and education after distal gastrectomy for gastric cancer patients to prevent loss of lean body mass in Korea: a non-randomized controlled study
-
Hee Kyung Yoon1
 , Sun Ae Kim2
, Sun Ae Kim2 , Ji Yoon Han3
, Ji Yoon Han3 , Dong Jin Kim1
, Dong Jin Kim1
-
Annals of Clinical Nutrition and Metabolism 2024;16(1):10-19.
DOI: https://doi.org/10.15747/ACNM.2024.16.1.10
Published online: April 1, 2024
1Department of Surgery, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
2Department of Nursing, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
3Department of Clinical Nutrition, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
- Corresponding author: Dong Jin Kim, email: djdjcap@catholic.ac.kr
© 2024 The Korean Society of Surgical Metabolism and Nutrition · The Korean Society for Parenteral and Enteral Nutrition
This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 5,870 Views
- 45 Download
Abstract
-
Purpose We studied whether active education of patients about the importance of a high-protein diet can prevent lean body mass loss after gastrectomy for gastric cancer.
-
Methods In the study group, intensive high protein diet education and monitoring was performed immediate post operative, 1, 3, and 6 months after surgery. Study group patients were compared with data from the control group formed using propensity matching with the study group for age, sex, resection extent, and TNM stage. Clinicopathologic factors were compared between the groups, and changes in quality of life (QOL) and lean body mass between preoperative levels and 6 months after surgery were assessed.
-
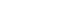
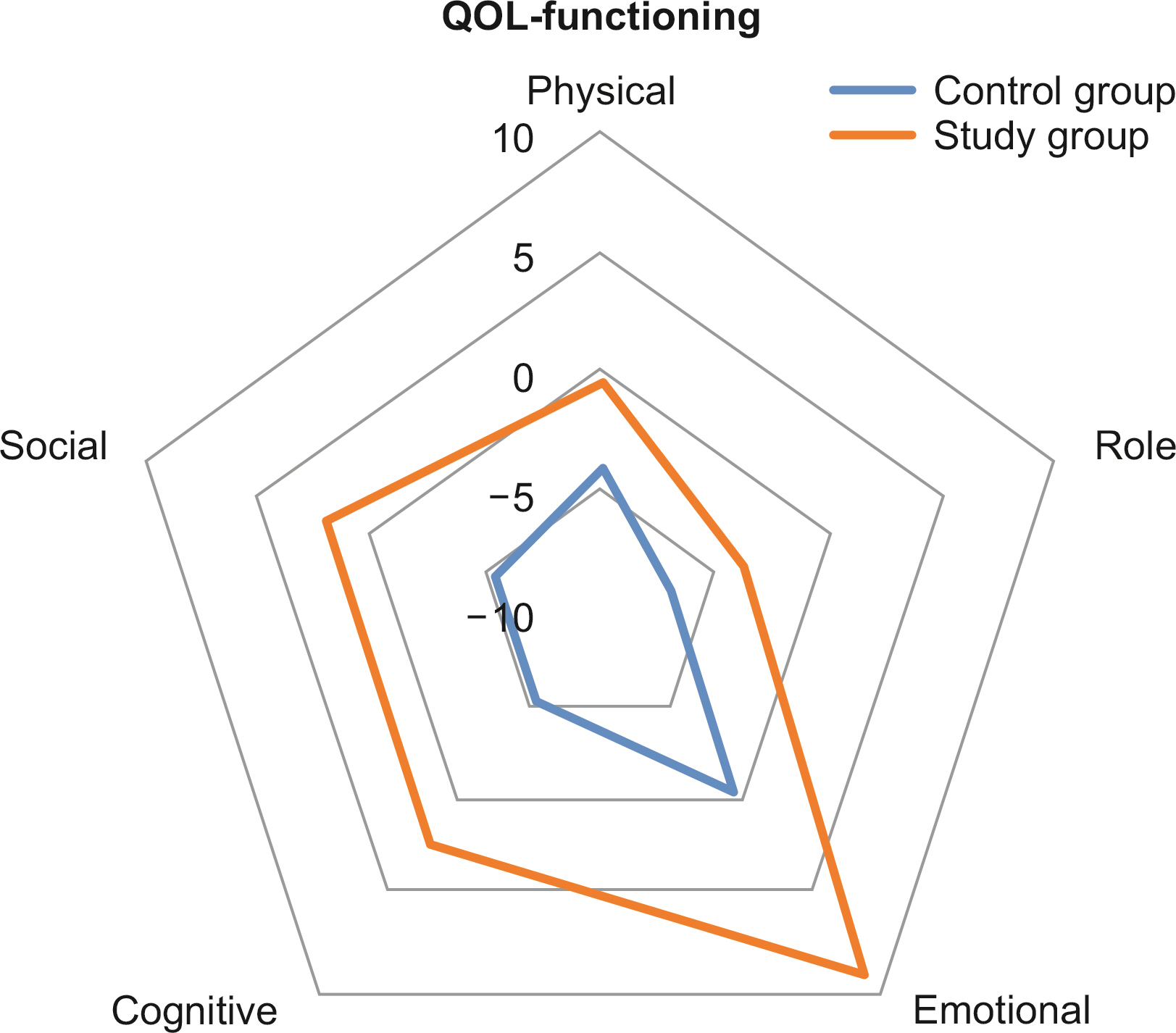
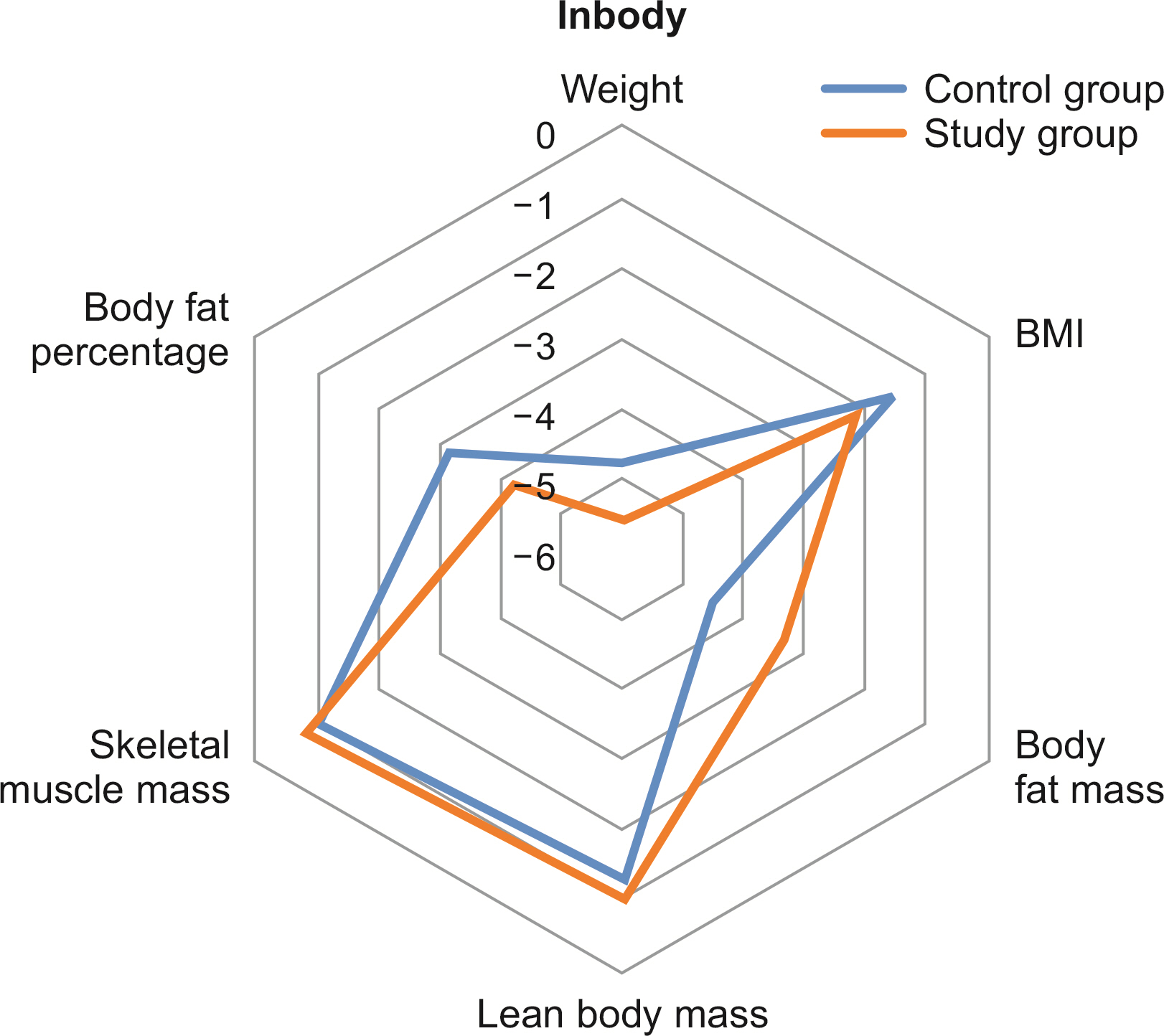
Results Among the 100 patients, 31 patients from each group were matched with propensity matching. The groups had no significant clinicopathologic differences. Although the changes in QOL scale and body composition did not differ statistically between the groups, a favorable trend was observed in the study group. Six months after surgery, the mean change in the QOL scale, which measured physical, role, emotional, cognitive, and social functioning, decreased less than the control group or even increased in the study group. In the body composition analysis, the study group showed greater reductions in weight, body mass index, fat mass, and body fat percentage than the control group, and their lean body mass and skeletal muscle mass decreased less.
-
Conclusion A high-protein diet protocol and education might increase patient QOL and prevent a decrease in lean body weight 6 months after distal gastric resection.
Introduction
Methods
Results
Discussion
Supplementary materials
Acknowledgments
Authors’ contribution
Conceptualization: DJK. Data curation: DJK, JYH. Formal analysis: DJK. Funding acquisition: DJK. Investigation: DJK. Methodology: SAK. Project administration: JYH, SAK. Resources: DJK. Software: DJK. Supervision: DJK. Validation: DJK, JYH. Visualization: HKY. Writing – original draft: HKY, DJK. Writing – review & editing: all authors.
Conflict of interest
The authors of this manuscript have no conflicts of interest to disclose.
Funding
This study was conducted with funds from the Korean Society of Surgical Metabolism and Nutrition (No.: KSSMN 2020-005).
Data availability
Raw data is not available due to institutional principle.
| Variable | Control group (n=31) | Within P-value | Study group (n=31) | Within P-value |
Between P-value |
Compliance (>90%) (n=22) |
Within P-valuea |
|---|---|---|---|---|---|---|---|
| Height (cm) | |||||||
| Preoperation | 163.35±8.39 | NA | 165.53±9.06 | >0.9999 (S) | 0.3332 (W) | 163.03±9.30 | |
| Weight (kg) | |||||||
| Preoperation | 67.94±12.33 | 65.43±11.46 | 0.4098 (T) | 63.85±9.67 | |||
| 6 months | 63.19±11.38 | 59.86±10.14 | 0.2292 (T) | 59.14±8.69 | |||
| Change at 6 months | –4.75±4.85 | <0.0001 (P) | –5.56±3.71 | <0.0001 (P) | 0.4596 (T) | –4.72±3.01 | <0.0001 |
| Body mass index (kg/m2) | |||||||
| Preoperation | 25.10±4.09 | 24.35±2.92 | 0.6676 (W) | 23.97±2.59 | |||
| 6 months | 23.65±3.82 | 22.19±2.49 | 0.1147 (W) | 22.07±2.25 | |||
| Change at 6 months | –1.45±2.07 | <0.0001 (S) | –2.17±1.36 | <0.0001 (P) | 0.1764 (W) | –1.90±1.11 | <0.0001 |
| Lean body mass (kg) | |||||||
| Preoperation | 48.27±9.01 | 47.12±9.50 | 0.6242 (T) | 46.29±9.08 | |||
| 6 months | 46.93±9.08 | 46.09±9.11 | 0.7178 (T) | 45.55±8.68 | |||
| Change at 6 months | –1.35±2.48 | 0.0001 (S) | –1.03±2.95 | 0.0029 (S) | 0.9775 (W) | –0.73±3.28 | 0.0428 |
| Body fat mass (kg) | |||||||
| Preoperation | 19.66±7.65 | 18.31±5.90 | 0.6986 (W) | 17.57±5.50 | |||
| 6 months | 16.26±7.13 | 13.78±4.59 | 0.2482 (W) | 13.58±4.73 | |||
| Change at 6 months | –3.40±3.69 | <0.0001 (P) | –4.53±3.37 | <0.0001 (P) | 0.2110 (T) | –3.99±3.00 | <0.0001 |
| Skeletal muscle mass (kg) | |||||||
| Preoperation | 45.59±8.56 | 44.52±9.01 | 0.6341 (T) | 43.75±8.60 | |||
| 6 months | 44.29±8.62 | 43.50±8.66 | 0.7201 (T) | 43.00±8.43 | |||
| Change at 6 months | –1.30±2.37 | 0.0002 (S) | –1.02±2.79 | 0.0026 (S) | 0.9719 (W) | –0.74±3.10 | 0.0375 |
| Body fat ratio (%) | |||||||
| Preoperation | 28.63±7.72 | 28.00±7.93 | 0.7527 (T) | 27.60±8.39 | |||
| 6 months | 25.47±8.39 | 23.15±7.29 | 0.2501 (T) | 23.19±8.28 | |||
| Change at 6 months | –3.15±4.16 | 0.0002 (P) | –4.18±4.47 | <0.0001 (P) | 0.1286 (T) | –4.41±4.52 | 0.0002 |
Values are presented as mean±standard deviation.
Between P-value.
Continuous data: independent t-test (T) or Wilcoxon’s rank-sum test (W).
Within P-value: paired t-test (P) or Wilcoxon’s signed-rank test (S).
NA = not applicable.
aWithin P-value = comparison before and after surgery among patients with high compliance.
- 1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71:209-49. ArticlePubMedPDF
- 2. Kang MJ, Jung KW, Bang SH, Choi SH, Park EH, Yun EH, et al. Community of Population-Based Regional Cancer Registries. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2020. Cancer Res Treat 2023;55:385-99. ArticlePubMedPMCPDF
- 3. Park SH, Kang MJ, Yun EH, Jung KW. Epidemiology of gastric cancer in Korea: trends in incidence and survival based on Korea Central Cancer Registry data (1999-2019). J Gastric Cancer 2022;22:160-8. ArticlePubMedPMCPDF
- 4. Information Committee of the Korean Gastric Cancer Association. Korean Gastric Cancer Association-led nationwide survey on surgically treated gastric cancers in 2019. J Gastric Cancer 2021;21:221-35. ArticlePubMedPMCPDF
- 5. Kim TH, Kim IH, Kang SJ, Choi M, Kim BH, Eom BW, et al. Development Working Groups for the Korean Practice Guidelines for Gastric Cancer 2022 Task Force Team. Korean practice guidelines for gastric cancer 2022: an evidence-based, multidisciplinary approach. J Gastric Cancer 2023;23:3-106. PubMedPMC
- 6. Mechanick JI, Apovian C, Brethauer S, Garvey WT, Joffe AM, Kim J, et al. Clinical practice guidelines for the perioperative nutrition, metabolic, and nonsurgical support of patients undergoing bariatric procedures - 2019 update: cosponsored by American Association of Clinical Endocrinologists/American College of Endocrinology, The Obesity Society, American Society for Metabolic & Bariatric Surgery, Obesity Medicine Association, and American Society of Anesthesiologists. Surg Obes Relat Dis 2020;16:175-247. ArticlePubMed
- 7. Bae JM, Kim S, Kim YW, Ryu KW, Lee JH, Noh JH, et al. Health-related quality of life among disease-free stomach cancer survivors in Korea. Qual Life Res 2006;15:1587-96. ArticlePubMedPDF
- 8. Lee SJ, Kim JY, Ha TK, Choi YY. Changes in lipid indices and body composition one year after laparoscopic gastrectomy: a prospective study. Lipids Health Dis 2018;17:113.ArticlePubMedPMCPDF
- 9. Li Y, Li S, Wang W, Zhang D. Association between dietary protein intake and cognitive function in adults aged 60 years and older. J Nutr Health Aging 2020;24:223-9. ArticlePubMedPMCPDF
- 10. Lim HS, Lee B, Cho I, Cho GS. Nutritional and clinical factors affecting weight and fat-free mass loss after gastrectomy in patients with gastric cancer. Nutrients 2020;12:1905.ArticlePubMedPMC
- 11. McLester CN, Nickerson BS, Kliszczewicz BM, McLester JR. Reliability and agreement of various InBody body composition analyzers as compared to dual-energy X-ray absorptiometry in healthy men and women. J Clin Densitom 2020;23:443-50. ArticlePubMed
- 12. Han SH, Lee SY, Kim KN, Yoo S. Comparison between Dual Energy X-ray Absorptiometry (DEXA) and Bioelectrical Impedence Analysis (BIA) in measurement of lean body mass and fat mass in obese adults -based bland-Altman Plot Analysis-. Korean J Obes 2006;15:213-8.
- 13. Cho YH, Choi SK, Kim DY, Woo JT, Kim SW, Yang IM, et al. Age-related difference in body fatness by bioelectrical impedance method and Dual-Energy X-ray Absorptiometry. J Korean Soc Stud Obes 1997;6:59-66.
- 14. Cho YG, Kang JH, Kim KA. Comparison of bioelectrical impedance analysis with Dual Energy X-ray Absorptiometry in obese women. Korean J Obes 2007;16:50-7.
References
Figure & Data
REFERENCES
Citations

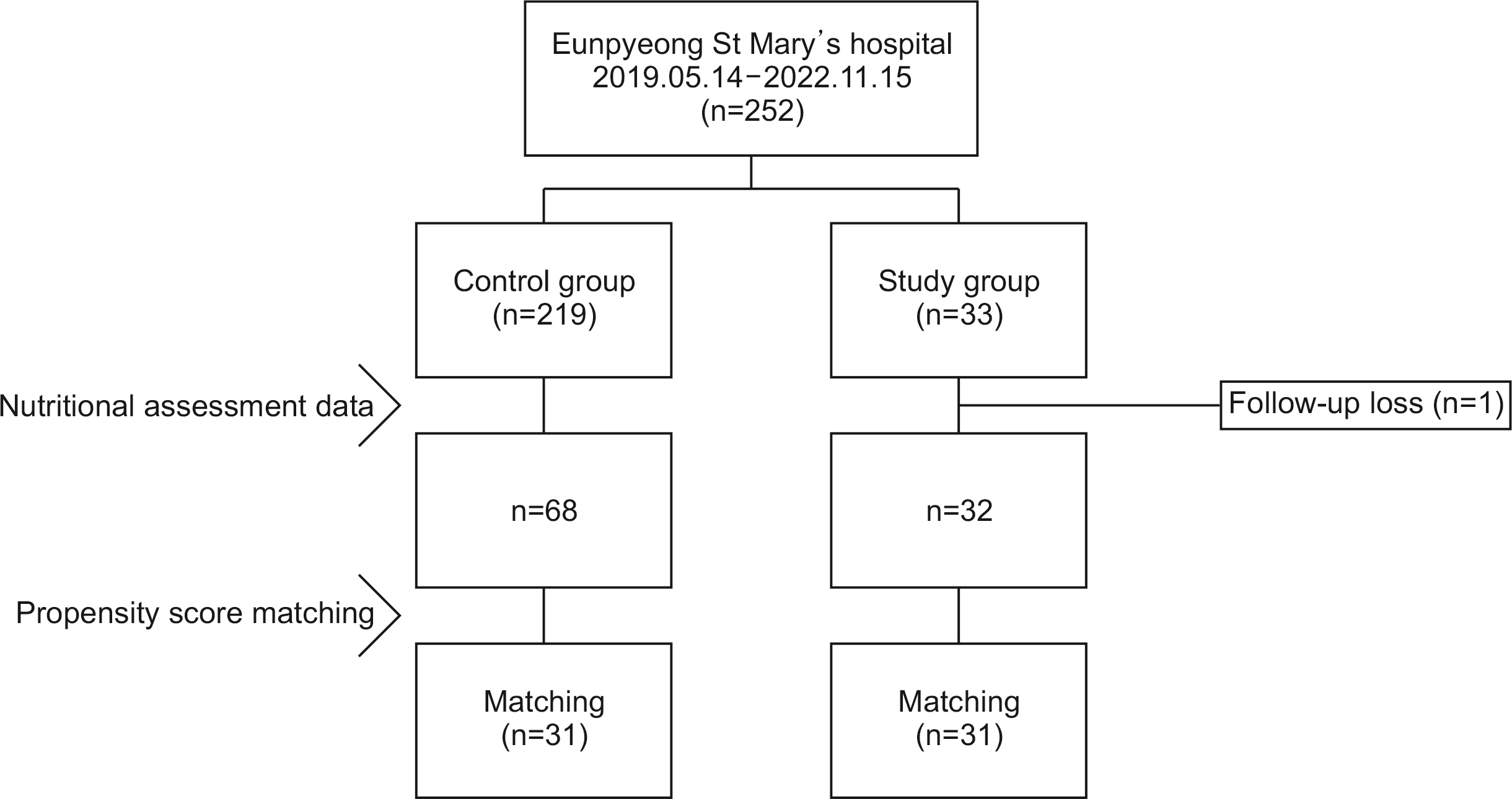
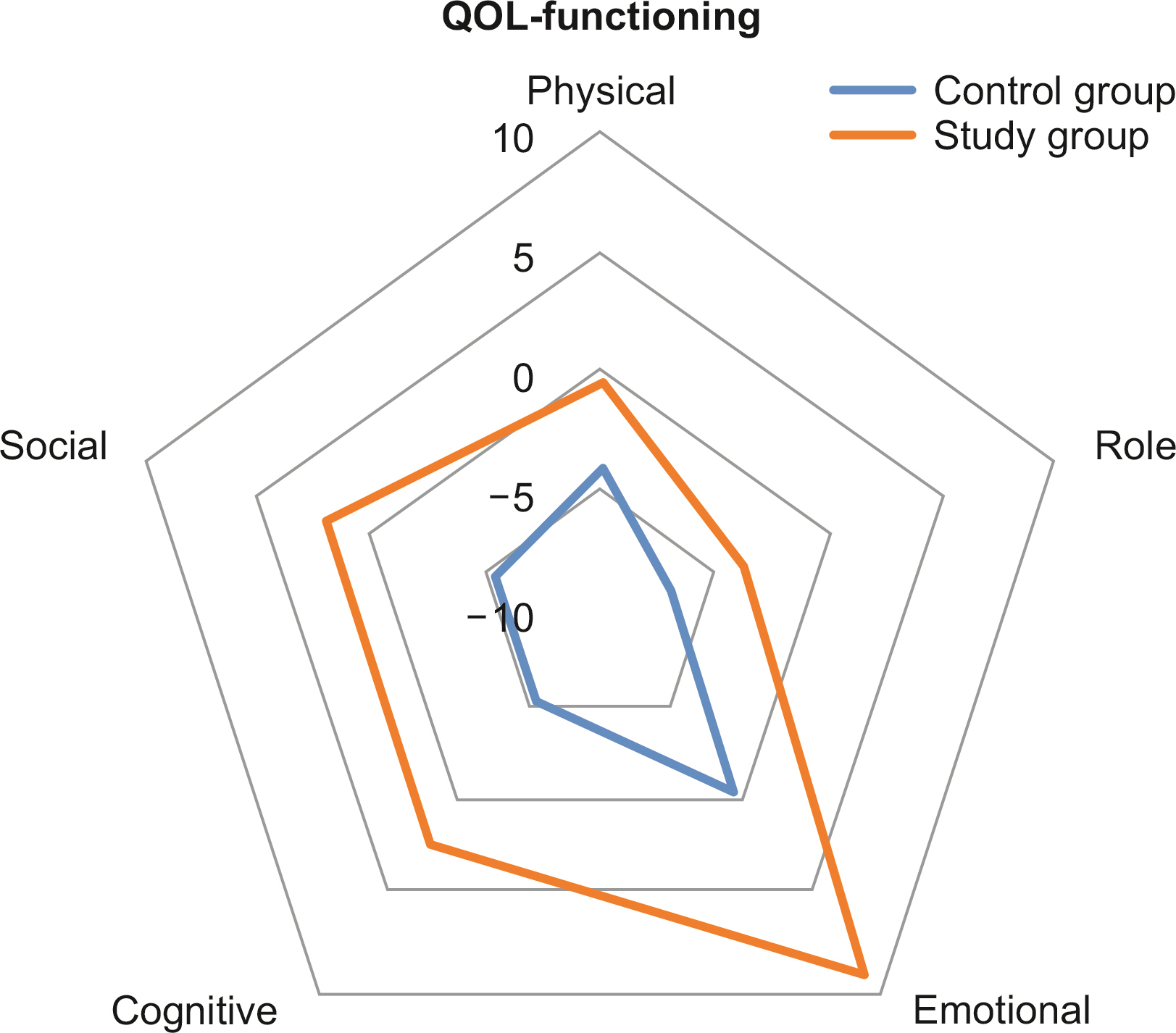
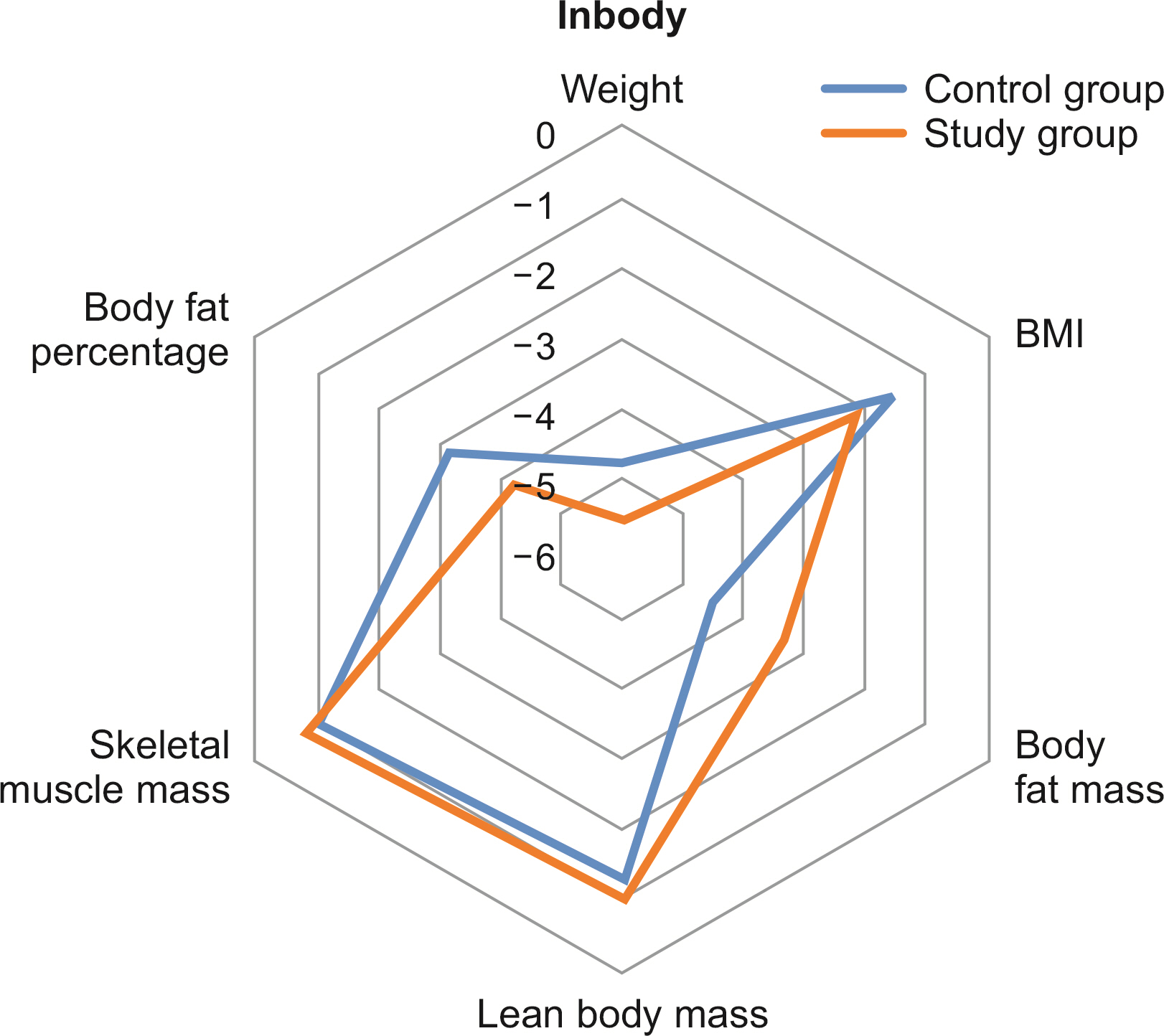
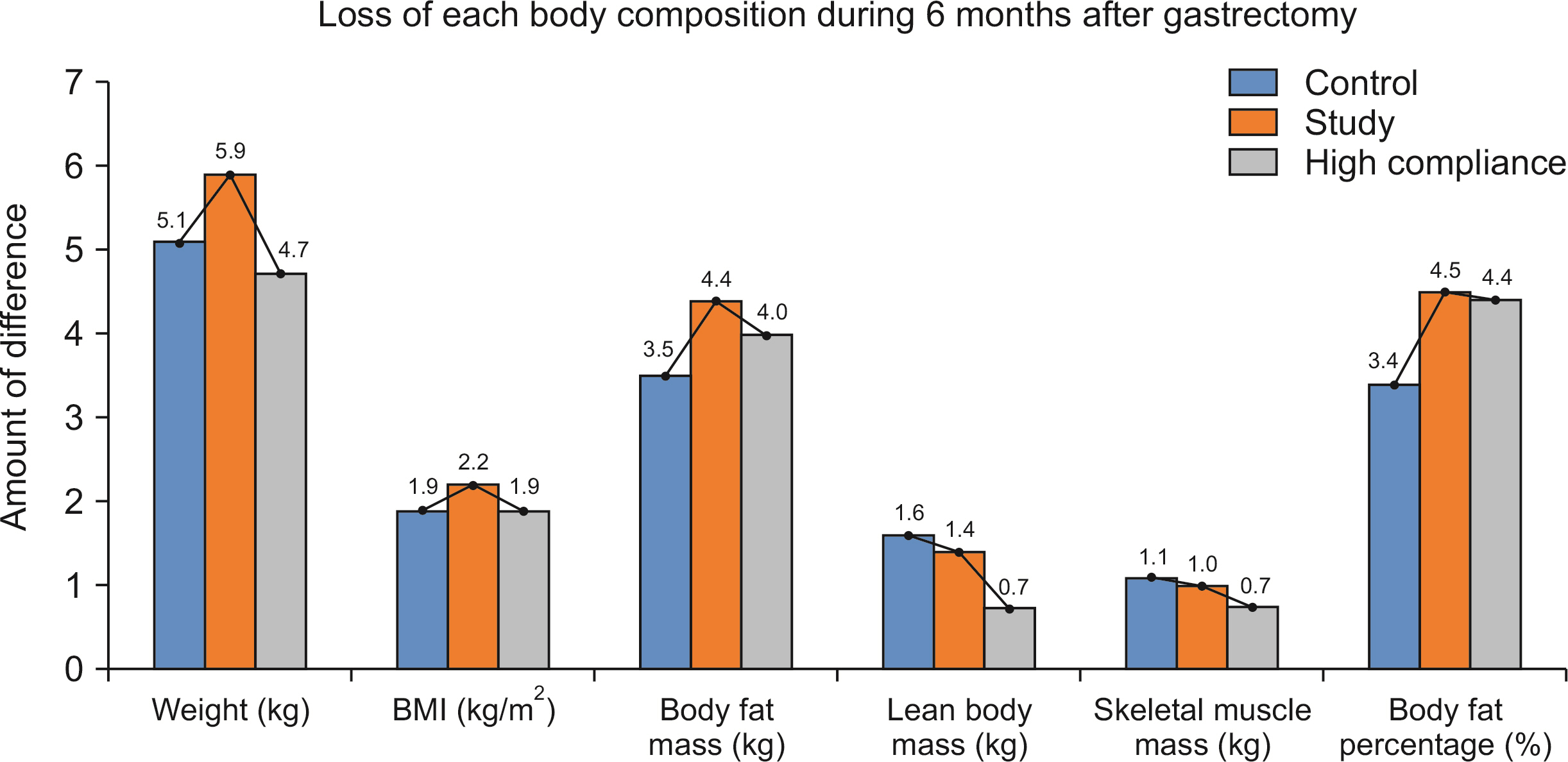
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Clinicopathological characteristic analysis between the control group and the study group
| Variable | Control group (n=31) | Study group (n=31) | P-value |
|---|---|---|---|
| Age (yr) | 64.10±8.73 | 62.48±9.07 | 0.5918 |
| Sex | |||
| Male | 20 (64.52) | 21 (67.74) | |
| Female | 11 (35.48) | 10 (32.26) | |
| ASA | |||
| 1 | 7 (22.58) | 4 (12.90) | 0.2975 |
| 2 | 19 (61.29) | 24 (77.42) | |
| 3 | 5 (16.13) | 2 (6.45) | |
| 4 | 0 | 1 (3.23) | |
| Body mass index (kg/m2) | 25.52±4.15 | 24.18±3.03 | 0.2846 |
| Smoking | |||
| Non-smoker | 13 (41.94) | 11 (35.48) | 0.8678 |
| Ex-smoker | 6 (19.35) | 7 (22.58) | |
| Current smoker | 12 (38.71) | 13 (41.94) | |
| Alcohol | |||
| Non-drinker | 13 (41.94) | 14 (45.16) | 0.7649 |
| Social-drinker | 14 (45.16) | 15 (48.39) | |
| Heavy-alcoholics | 4 (12.90) | 2 (6.45) | |
| Comorbidity | |||
| None | 11 (35.48) | 14 (45.16) | 0.7761 |
| Yes | 20 (64.52) | 17 (54.84) | |
| Approach | |||
| Open | 2 (6.45) | 0 | 0.2067 |
| Lap assist | 0 | 0 | |
| Totally lap | 28 (90.32) | 31 (100.0) | |
| Open conversion | 1 (3.23) | 0 | |
| Operation time (min) | 211.68±62.42 | 198.97±43.40 | 0.3557 |
| Estimated blood loss (mL) | 65.16±100.54 | 50.97±60.35 | 0.3970 |
| Hospital day | 8.67±3.27 | 9.07±4.29 | 0.9868 |
| Extent of resection | |||
| Distal gastrectomy | 31 (100.0) | 31 (100.0) | NA |
| Combined resection | |||
| None | 31 (100.0) | 31 (100.0) | NA |
| Lymph node dissection | |||
| Below D1+ | 14 (45.17) | 13 (41.94) | 0.3515 |
| Above D2 | 17 (54.84) | 18 (58.06) | |
| Reconstruction | |||
| Billroth-I | 6 (19.35) | 8 (25.81) | 0.5134 |
| Billroth-II | 23 (74.19) | 19 (61.29) | |
| Roux-en-Y | 2 (6.45) | 4 (12.90) | |
| Complex combined resection | 0 | 0 | |
| Size | 3.30±2.48 | 3.19±1.69 | 0.8711 |
| Number | |||
| 1 | 29 (93.55) | 29 (93.55) | >0.9999 |
| 2 | 1 (3.23) | 2 (6.45) | |
| 3 | 1 (3.23) | 0 | |
| Location | |||
| Middle 1/3 | 12 (38.71) | 14 (45.16) | 0.4467 |
| Lower 1/3 | 19 (61.29) | 17 (54.85) | |
| Differentiation | |||
| W-tubular | 6 (19.35) | 9 (29.03) | 0.8443 |
| M-tubular | 10 (32.26) | 10 (32.26) | |
| P-tubular | 6 (19.35) | 5 (16.13) | |
| Poorly cohesive | 9 (29.03) | 7 (22.58) | |
| p.Stage.8th | |||
| Ia | 18 (58.06) | 18 (58.06) | 0.1346 |
| Ib | 3 (9.68) | 3 (9.68) | |
| IIa | 6 (19.35) | 1 (3.23) | |
| IIb | 3 (9.68) | 2 (6.45) | |
| IIIa | 1 (3.23) | 1 (3.23) | |
| IIIb | 0 | 4 (12.90) | |
| IIIc | 0 | 2 (6.45) |
Values are presented as mean±standard deviation or number (%).
ASA = American Society of Anesthesiologists, physical status classification system; W-tubular = well-differentiated tubular; M-tubular = moderately differentiated tubular; P-tubular = poorly differentiated tubular; NA = not applicable.
Quality of life questionnaire European Organization for Research and Treatment Center Quality of Life Questionnaire (EORTC QLQ)-C30 analysis between the control group and study group
| Variable | Control group (n=31) | Within P-value |
Study group (n=31) | Within P-value |
Between P-value |
|---|---|---|---|---|---|
| Global health status | |||||
| Preoperation | 69.89±20.83 | 63.44±22.12 | 0.2266 | ||
| 6 months | 68.06±18.84 | 65.05±20.57 | 0.6433 | ||
| Change at 6 months | –1.11±22.18 | 0.7291 (S) | 1.61±29.06 | 0.7594 (P) | 0.7926 (W) |
| 1. Physical functioning | |||||
| Preoperation | 87.10±12.04 | 84.95±13.66 | 0.4815 | ||
| 6 months | 83.01±10.45 | 84.52±15.14 | 0.2300 | ||
| Change at 6 months | –4.09±14.47 | 0.1264 (P) | –0.43±13.21 | 0.9463 (S) | 0.1813 (W) |
| 2. Role functioning | |||||
| Preoperation | 95.16±13.05 | 87.10±17.59 | 0.0311 | ||
| 6 months | 88.17±14.40 | 83.33±21.94 | 0.5070 | ||
| Change at 6 months | –6.99±14.77 | 0.0059 (S) | –3.76±25.35 | 0.7336 (S) | 0.1861 (W) |
| 3. Emotional functioning | |||||
| Preoperation | 85.75±17.37 | 79.30±16.08 | 0.0851 | ||
| 6 months | 85.22±20.04 | 87.37±14.09 | 0.9122 | ||
| Change at 6 months | –0.54±17.07 | 0.7648 (S) | 8.06±17.01 | 0.0101 (S) | 0.2148 (W) |
| 4. Cognitive functioning | |||||
| Preoperation | 88.71±10.88 | 84.95±12.44 | 0.2426 | ||
| 6 months | 83.33±14.91 | 87.10±9.34 | 0.4095 | ||
| Change at 6 months | –5.38±16.32 | 0.1170 (S) | 2.15±14.75 | 0.7227 (S) | 0.0905 (W) |
| 5. Social functioning | |||||
| Preoperation | 87.10±22.65 | 87.63±21.93 | 0.9665 | ||
| 6 months | 81.72±18.93 | 889.78±19.09 | 0.3070 | ||
| Change at 6 months | –5.38±25.96 | 0.3245 (S) | 2.15±26.44 | 0.9587 (S) | 0.1338 (W) |
Values are presented as mean±standard deviation.
Between P-value
Continuous data: Wilcoxon’s rank-sum test (W).
Within P-value: paired t-test (P) or Wilcoxon’s signed-rank test (S).
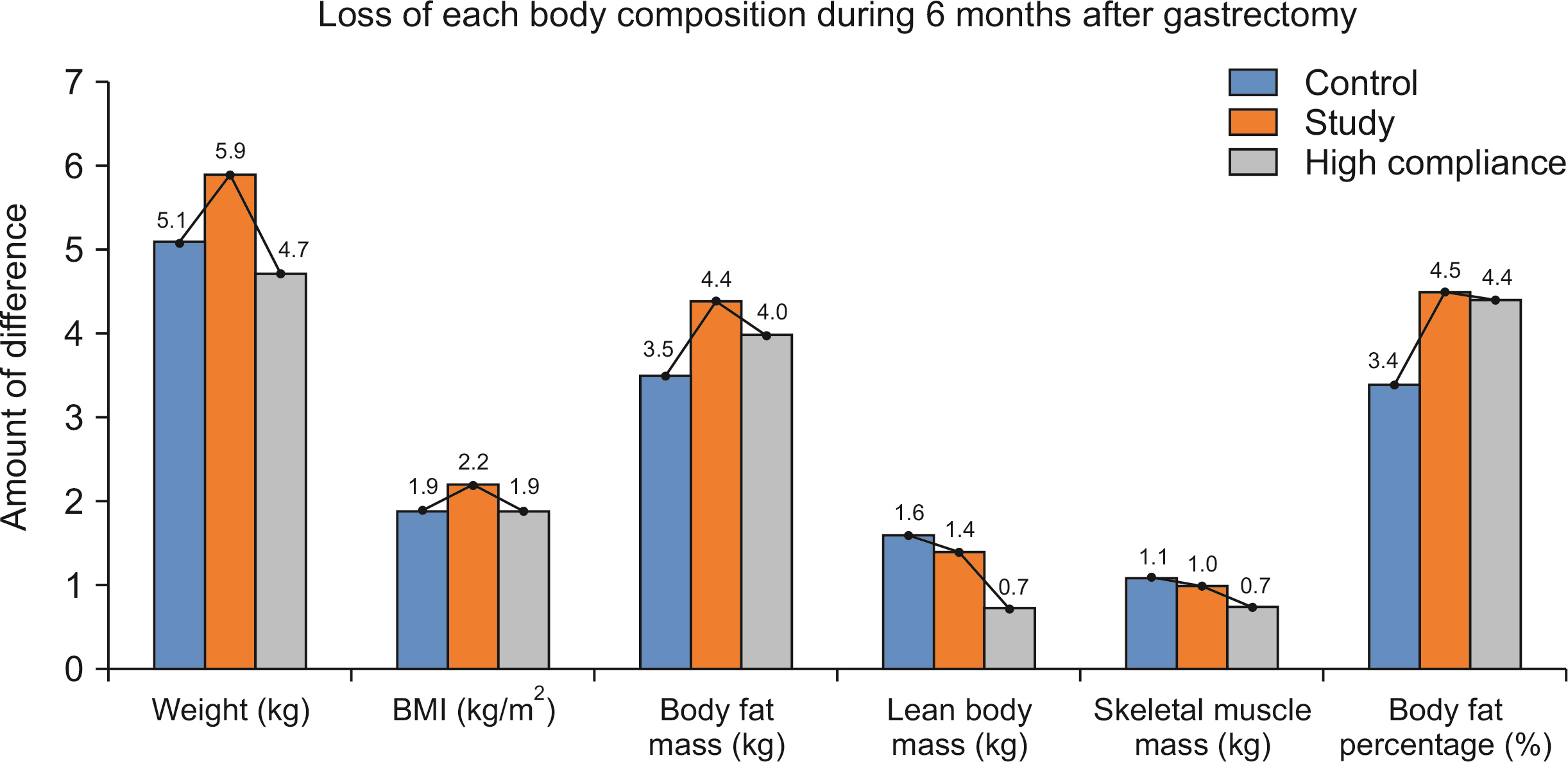
Body composition analysis between the control group and the study group
| Variable | Control group (n=31) | Within P-value | Study group (n=31) | Within P-value | Between P-value |
Compliance (>90%) (n=22) |
Within P-value |
|---|---|---|---|---|---|---|---|
| Height (cm) | |||||||
| Preoperation | 163.35±8.39 | NA | 165.53±9.06 | >0.9999 (S) | 0.3332 (W) | 163.03±9.30 | |
| Weight (kg) | |||||||
| Preoperation | 67.94±12.33 | 65.43±11.46 | 0.4098 (T) | 63.85±9.67 | |||
| 6 months | 63.19±11.38 | 59.86±10.14 | 0.2292 (T) | 59.14±8.69 | |||
| Change at 6 months | –4.75±4.85 | <0.0001 (P) | –5.56±3.71 | <0.0001 (P) | 0.4596 (T) | –4.72±3.01 | <0.0001 |
| Body mass index (kg/m2) | |||||||
| Preoperation | 25.10±4.09 | 24.35±2.92 | 0.6676 (W) | 23.97±2.59 | |||
| 6 months | 23.65±3.82 | 22.19±2.49 | 0.1147 (W) | 22.07±2.25 | |||
| Change at 6 months | –1.45±2.07 | <0.0001 (S) | –2.17±1.36 | <0.0001 (P) | 0.1764 (W) | –1.90±1.11 | <0.0001 |
| Lean body mass (kg) | |||||||
| Preoperation | 48.27±9.01 | 47.12±9.50 | 0.6242 (T) | 46.29±9.08 | |||
| 6 months | 46.93±9.08 | 46.09±9.11 | 0.7178 (T) | 45.55±8.68 | |||
| Change at 6 months | –1.35±2.48 | 0.0001 (S) | –1.03±2.95 | 0.0029 (S) | 0.9775 (W) | –0.73±3.28 | 0.0428 |
| Body fat mass (kg) | |||||||
| Preoperation | 19.66±7.65 | 18.31±5.90 | 0.6986 (W) | 17.57±5.50 | |||
| 6 months | 16.26±7.13 | 13.78±4.59 | 0.2482 (W) | 13.58±4.73 | |||
| Change at 6 months | –3.40±3.69 | <0.0001 (P) | –4.53±3.37 | <0.0001 (P) | 0.2110 (T) | –3.99±3.00 | <0.0001 |
| Skeletal muscle mass (kg) | |||||||
| Preoperation | 45.59±8.56 | 44.52±9.01 | 0.6341 (T) | 43.75±8.60 | |||
| 6 months | 44.29±8.62 | 43.50±8.66 | 0.7201 (T) | 43.00±8.43 | |||
| Change at 6 months | –1.30±2.37 | 0.0002 (S) | –1.02±2.79 | 0.0026 (S) | 0.9719 (W) | –0.74±3.10 | 0.0375 |
| Body fat ratio (%) | |||||||
| Preoperation | 28.63±7.72 | 28.00±7.93 | 0.7527 (T) | 27.60±8.39 | |||
| 6 months | 25.47±8.39 | 23.15±7.29 | 0.2501 (T) | 23.19±8.28 | |||
| Change at 6 months | –3.15±4.16 | 0.0002 (P) | –4.18±4.47 | <0.0001 (P) | 0.1286 (T) | –4.41±4.52 | 0.0002 |
Values are presented as mean±standard deviation.
Between P-value.
Continuous data: independent t-test (T) or Wilcoxon’s rank-sum test (W).
Within P-value: paired t-test (P) or Wilcoxon’s signed-rank test (S).
NA = not applicable.
aWithin P-value = comparison before and after surgery among patients with high compliance.
Values are presented as mean±standard deviation or number (%). ASA = American Society of Anesthesiologists, physical status classification system; W-tubular = well-differentiated tubular; M-tubular = moderately differentiated tubular; P-tubular = poorly differentiated tubular; NA = not applicable.
Values are presented as mean±standard deviation. Between P-value Continuous data: Wilcoxon’s rank-sum test (W). Within P-value: paired t-test (P) or Wilcoxon’s signed-rank test (S).
Values are presented as mean±standard deviation. Between P-value. Continuous data: independent t-test (T) or Wilcoxon’s rank-sum test (W). Within P-value: paired t-test (P) or Wilcoxon’s signed-rank test (S). NA = not applicable. aWithin P-value = comparison before and after surgery among patients with high compliance.

 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN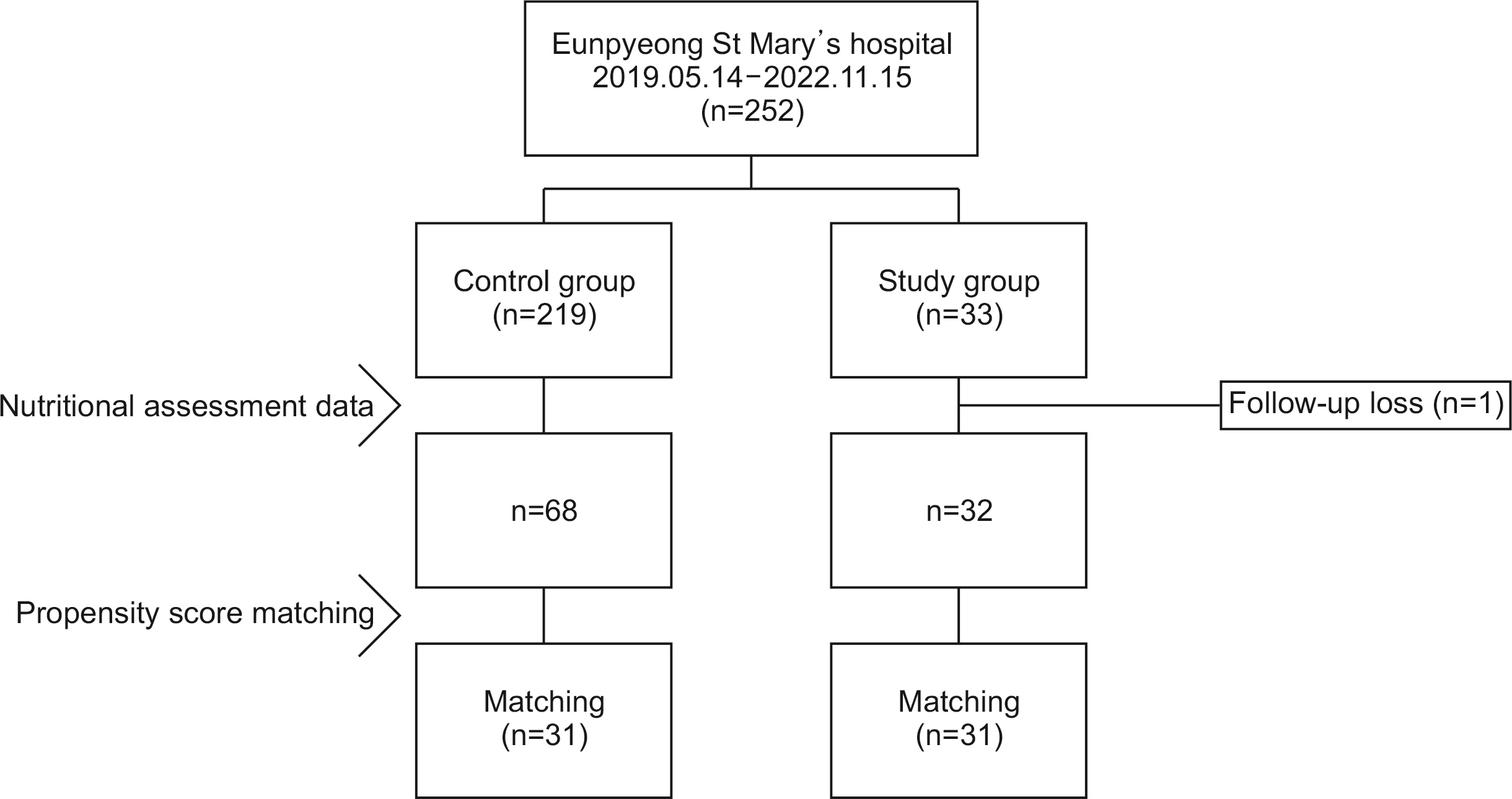
 Cite
Cite