Scopus, KCI, KoreaMed

Articles
- Page Path
- HOME > Surgical Metabolism and Nutrition > Volume 10(1); 2019 > Article
- ORIGINAL ARTICLE Physiological Change of Serum Bilirubin Level by ω-3 Enriched Parenteral Nutrition Versus ω-3 Free Parenteral Nutrition in Healthy Male Subjects
- Mi Rin Lee, M.D., Ph.D.1, Min-Gul Kim, M.D., Ph.D.2,3,4, Jae Do Yang, M.D., Ph.D.1,4, Hong Pil Hwang, M.D., Ph.D.1,4, Yunjeong Kim, Ph.D.3,4, Ji-Young Jeon, Ph.D.3,4, Sungwoo Ahn, M.D.1,4, Hee Chul Yu, M.D., Ph.D.1,4,5
-
Surgical Metabolism and Nutrition 2019;10(1):20-26.
DOI: https://doi.org/10.18858/smn.2019.10.1.20
Published online: June 30, 2019
Department of Surgery, Chonbuk National University Medical School, Jeonju, Korea
Department of Pharmacology, Chonbuk National University Medical School, Jeonju, Korea
Department of Center for Clinical Pharmacology, Chonbuk National University Hospital, Jeonju, Korea
Department of Research Institute of Clinical Medicine of Chonbuk National University-Biomedical Research Institute of Chonbuk University Hospital, Jeonju, Korea
Department of Research Institute for Endocrine Sciences, Chonbuk National University, Jeonju, Korea
Department of Surgery, Chonbuk National University Medical School, Jeonju, Korea
Department of Pharmacology, Chonbuk National University Medical School, Jeonju, Korea
Department of Center for Clinical Pharmacology, Chonbuk National University Hospital, Jeonju, Korea
Department of Research Institute of Clinical Medicine of Chonbuk National University-Biomedical Research Institute of Chonbuk University Hospital, Jeonju, Korea
Department of Research Institute for Endocrine Sciences, Chonbuk National University, Jeonju, Korea
- Correspondence to: Hee Chul Yu, Department of Surgery, Chonbuk National University Medical School, 20 Geonji-ro, Deokjin-gu, Jeonju 54907, Korea Tel: +82-63-250-1576, Fax: +82-63-271-6197, E-mail: hcyu@jbnu.ac.kr
This study was supported by JW Pharmaceutical, Corp., Seoul, Republic of Korea.
This article received the academic award in the 23th Fall Conference, Surgical metabolism and Nutrition, 2017.
Copyright: © The Korean Society of Surgical Metabolism and Nutrition
This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 2,714 Views
- 8 Download
Abstract
-
Purpose: Bilirubin is a biomarker for the diagnosis of liver diseases or bile duct dysfunction. This study assessed the physiological changes in the blood bilirubin level infusing ω-3 enriched parenteral nutrition (PN) and ω-3 free PN in healthy male subjects.
-
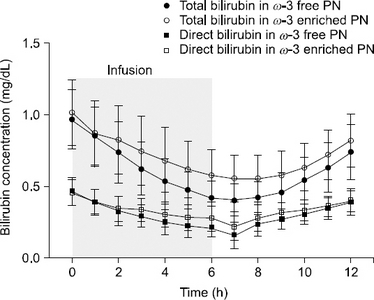
Materials and Methods: This study was a randomized, open-label, two-treatment, two-way crossover trial. Sixteen subjects were assigned randomly to one of two sequences of the two treatments: ω-3 enriched PN or ω-3 free PN was infused via aperipheral venous catheter for six hours at 3 mL/kg/h. Blood samples were collected every one hour from 0 to 12 hours after starting an intravenous infusion for bilirubin concentrations. The total bilirubin and direct bilirubin concentrations in the blood were analyzed using an enzymatic method.
-
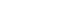
Results: The bilirubin concentration in the blood was reduced while infusing the ω-3 enriched PN and ω-3 free PN. When it stopped infusing, the bilirubin concentration was recovered. A similar pattern was observed, but there was a further decline and recovery in ω-3 free PN.
-
Conclusion: When ω-3 enriched PN and ω-3 free PN are infused in healthy male subjects, the blood bilirubin level decreasedand there is no difference between the two groups.
INTRODUCTION
MATERIALS AND METHODS
RESULTS
| Group A (n=8) | Group B (n=8) | Total (n=16) | P-value1) | |
|---|---|---|---|---|
| Ages (years) | 34.8±6.54 | 35.5±6.37 | 35.1±6.25 | 0.820 |
| Height (cm) | 170.8±6.58 | 172.6±2.98 | 171.7±5.01 | 0.504 |
| Weight (kg) | 69.1±6.00 | 74.1±9.24 | 71.6±7.97 | 0.214 |
| BMI (kg/m2) | 23.7±2.24 | 24.9±2.89 | 24.3±2.57 | 0.383 |
| Time | Total bilirubin (mg/dL) | P-value1) | Direct bilirubin (mg/dL) | P-value1) | ||
|---|---|---|---|---|---|---|
|
|
|
|||||
| ω-3 enriched parenteral nutrition | ω-3 free parenteral nutrition | ω-3 enriched parenteral nutrition | ω-3 free parenteral nutrition | |||
| 0 (Baseline) | 1.01±0.23 | 0.97±0.21 | - | 0.46±0.09 | 0.46±0.09 | - |
| 1 | 0.87±0.22* | 0.84±0.21 | 0.6409 | 0.39±0.09* | 0.39±0.09* | 0.5495 |
| 2 | 0.82±0.24* | 0.74±0.21* | 0.3938 | 0.34±0.09* | 0.32±0.10* | 0.2889 |
| 3 | 0.75±0.24* | 0.62±0.18* | 0.0911 | 0.34±0.08* | 0.29±0.08* | 0.0227 |
| 4 | 0.68±0.22* | 0.54±0.16* | 0.0335 | 0.30±0.09* | 0.25±0.09* | 0.0053 |
| 5 | 0.62±0.20* | 0.47±0.13* | 0.0394 | 0.28±0.09* | 0.22±0.08* | 0.0041 |
| 6 | 0.57±0.18* | 0.42±0.12* | 0.0194 | 0.27±0.09* | 0.21±0.06* | 0.0022 |
| 7 | 0.55±0.17* | 0.40±0.11* | 0.0303 | 0.22±0.10* | 0.16±0.09* | 0.0014 |
| 8 | 0.55±0.16* | 0.42±0.12* | 0.0684 | 0.28±0.09* | 0.24±0.08* | 0.0300 |
| 9 | 0.58±0.17* | 0.45±0.13* | 0.1045 | 0.31±0.08* | 0.27±0.07* | 0.0244 |
| 10 | 0.63±0.16* | 0.54±0.16* | 0.3242 | 0.33±0.07* | 0.30±0.07* | 0.1090 |
| 11 | 0.72±0.17* | 0.63±0.17* | 0.3721 | 0.36±0.07* | 0.34±0.07* | 0.2649 |
| 12 | 0.82±0.19* | 0.74±0.19* | 0.5576 | 0.40±0.08* | 0.39±0.09* | 0.5315 |
| Time | Total bilirubin (mg/dL) | P-value1) | Direct bilirubin (mg/dL) | P-value1) | ||
|---|---|---|---|---|---|---|
|
|
|
|||||
| ω-3 enriched parenteral nutrition | ω-3 free parenteral nutrition | ω-3 enriched parenteral nutrition | ω-3 free parenteral nutrition | |||
| Maximum reduction | 0.48±0.12 | 0.57± 0.13 | 0.0038 | 0.25±0.06 | 0.32±0.12 | 0.0060 |
DISCUSSION
CONCLUSION
STATEMENT OF AUTHORSHIP
CONFLICT OF INTEREST
- 1. Wretlind A. Development of fat emulsions. JPEN J Parenter Enteral Nutr 1981;5:230-5. PubMed
- 2. Adolph M, Heller AR, Koch T, Koletzko B, Kreymann KG, Krohn K, et al. Working group for developing the guidelines for parenteral nutrition of The German Association for Nutritional Medicine. Lipid emulsions-guidelines on parenteral nutrition, chapter 6. Ger Med Sci 2009;7:Doc22.PubMedPMC
- 3. Piper SN, Schade I, Beschmann RB, Maleck WH, Boldt J, Röhm KD. Hepatocellular integrity after parenteral nutrition: comparison of a fish-oil-containing lipid emulsion with an olive-soybean oil-based lipid emulsion. Eur J Anaesthesiol 2009;26:1076-82. ArticlePubMed
- 4. Nanji AA, Anderson FH. Sensitivity and specificity of liver function tests in the detection of parenteral nutrition-associated cholestasis. JPEN J Parenter Enteral Nutr 1985;9:307-8. ArticlePubMed
- 5. Smithers LG, Gibson RA, McPhee A, Makrides M. Effect of long-chain polyunsaturated fatty acid supplementation of pre-term infants on disease risk and neurodevelopment: a systematic review of randomized controlled trials. Am J Clin Nutr 2008;87:912-20. ArticlePubMed
- 6. Clayton PT, Bowron A, Mills KA, Massoud A, Casteels M, Milla PJ. Phytosterolemia in children with parenteral nutrition-associated cholestatic liver disease. Gastroenterology 1993;105:1806-13. ArticlePubMed
- 7. Le HD, de Meijer VE, Robinson EM, Zurakowski D, Potemkin AK, Arsenault DA, et al. Parenteral fish-oil-based lipid emulsion improves fatty acid profiles and lipids in parenteral nutrition-dependent children. Am J Clin Nutr 2011;94:749-58. ArticlePubMed
- 8. Triana Junco M, García Vázquez N, Zozaya C, Ybarra Zabala M, Abrams S, García de Lorenzo A, et al. An exclusively based parenteral fish-oil emulsion reverses cholestasis. Nutr Hosp 2014;31:514-6. PubMed
- 9. Brans YW, Ritter DA, Kenny JD, Andrew DS, Dutton EB, Carrillo DW. Influence of intravenous fat emulsion on serum bilirubin in very low birthweight neonates. Arch Dis Child 1987;62:156-60. ArticlePubMedPMC
- 10. Andrew G, Chan G, Schiff D. Lipid metabolism in the neonate. II. The effect of Intralipid on bilirubin binding in vitro and in vivo. J Pediatr 1976;88:279-84. PubMed
- 11. Osawa S, Sugo S, Yoshida T, Yamaoka T, Nomura F. An assay for separating and quantifying four bilirubin fractions in un-treated human serum using isocratic high-performance liquid chromatography. Clin Chim Acta 2006;366:146-55. ArticlePubMed
- 12. Morimoto Y, Ishihara T, Takayama M, Kaito M, Adachi Y. Novel assay for measuring serum conjugated bilirubin and its clinical relevance. J Clin Lab Anal 2000;14:27-31. ArticlePubMedPMC
References
Figure & Data
REFERENCES
Citations

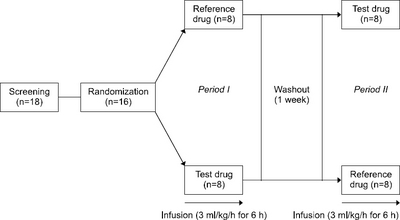
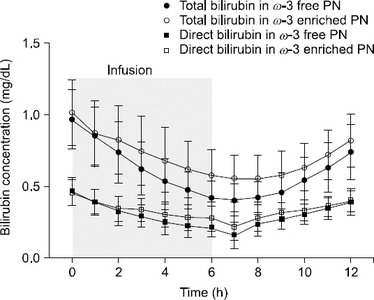
Fig. 1
Fig. 2
Demographic characteristics of study subjects (mean±SD)
| Group A (n=8) | Group B (n=8) | Total (n=16) | P-value |
|
|---|---|---|---|---|
| Ages (years) | 34.8±6.54 | 35.5±6.37 | 35.1±6.25 | 0.820 |
| Height (cm) | 170.8±6.58 | 172.6±2.98 | 171.7±5.01 | 0.504 |
| Weight (kg) | 69.1±6.00 | 74.1±9.24 | 71.6±7.97 | 0.214 |
| BMI (kg/m2) | 23.7±2.24 | 24.9±2.89 | 24.3±2.57 | 0.383 |
1)independent t-test to test the hypothesis that the difference between the means of two groups is equal to 0.
Total bilirubin and direct bilirubin concentration in ω-3 enriched parenteral nutrition and ω-3 free parenteral nutrition (mean±SD)
| Time | Total bilirubin (mg/dL) | P-value |
Direct bilirubin (mg/dL) | P-value |
||
|---|---|---|---|---|---|---|
| ω-3 enriched parenteral nutrition | ω-3 free parenteral nutrition | ω-3 enriched parenteral nutrition | ω-3 free parenteral nutrition | |||
| 0 (Baseline) | 1.01±0.23 | 0.97±0.21 | - | 0.46±0.09 | 0.46±0.09 | - |
| 1 | 0.87±0.22 |
0.84±0.21 | 0.6409 | 0.39±0.09 |
0.39±0.09 |
0.5495 |
| 2 | 0.82±0.24 |
0.74±0.21 |
0.3938 | 0.34±0.09 |
0.32±0.10 |
0.2889 |
| 3 | 0.75±0.24 |
0.62±0.18 |
0.0911 | 0.34±0.08 |
0.29±0.08 |
0.0227 |
| 4 | 0.68±0.22 |
0.54±0.16 |
0.0335 | 0.30±0.09 |
0.25±0.09 |
0.0053 |
| 5 | 0.62±0.20 |
0.47±0.13 |
0.0394 | 0.28±0.09 |
0.22±0.08 |
0.0041 |
| 6 | 0.57±0.18 |
0.42±0.12 |
0.0194 | 0.27±0.09 |
0.21±0.06 |
0.0022 |
| 7 | 0.55±0.17 |
0.40±0.11 |
0.0303 | 0.22±0.10 |
0.16±0.09 |
0.0014 |
| 8 | 0.55±0.16 |
0.42±0.12 |
0.0684 | 0.28±0.09 |
0.24±0.08 |
0.0300 |
| 9 | 0.58±0.17 |
0.45±0.13 |
0.1045 | 0.31±0.08 |
0.27±0.07 |
0.0244 |
| 10 | 0.63±0.16 |
0.54±0.16 |
0.3242 | 0.33±0.07 |
0.30±0.07 |
0.1090 |
| 11 | 0.72±0.17 |
0.63±0.17 |
0.3721 | 0.36±0.07 |
0.34±0.07 |
0.2649 |
| 12 | 0.82±0.19 |
0.74±0.19 |
0.5576 | 0.40±0.08 |
0.39±0.09 |
0.5315 |
1)General linear mixed model with Sequence, Treatment, Time, Treatment.
*Time effect as fixed effect and subject effect as random effect.
Maximum reduction of total bilirubin and direct bilirubin compared to baseline (mean±SD)
| Time | Total bilirubin (mg/dL) | P-value |
Direct bilirubin (mg/dL) | P-value |
||
|---|---|---|---|---|---|---|
| ω-3 enriched parenteral nutrition | ω-3 free parenteral nutrition | ω-3 enriched parenteral nutrition | ω-3 free parenteral nutrition | |||
| Maximum reduction | 0.48±0.12 | 0.57± 0.13 | 0.0038 | 0.25±0.06 | 0.32±0.12 | 0.0060 |
1)General linear mixed model with Sequence, Treatment, Period effect as fixed effect and sequence nested subject effect as random effect.
independent t-test to test the hypothesis that the difference between the means of two groups is equal to 0.
General linear mixed model with Sequence, Treatment, Time, Treatment. Time effect as fixed effect and subject effect as random effect.
General linear mixed model with Sequence, Treatment, Period effect as fixed effect and sequence nested subject effect as random effect.

 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN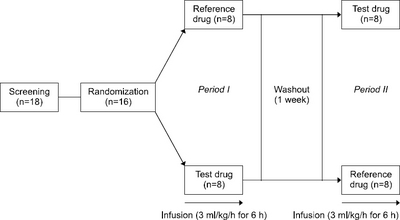
 Cite
Cite