Abstract
-
Purpose
This study investigated change of body mass index (BMI) and nutritional biochemical markers after gastrectomy according to the preoperative obesity in the elderly gastric cancer patients, and evaluated their short- and long-term clinical outcomes.
-
Materials and Methods
Gastric cancer patients (age≥65 years, BMI≥25 kg/m2) who underwent curative gastrectomy from 2014 to 2015 were enrolled in this study. BMI, comorbidity, extent of surgical resection, surgical approaches, postoperative complications, cancer stage, adjuvant chemotherapy, and survival data were collected. Nutritional parameters were collected until 4 years after surgery. The patients were classified into two groups according to preoperative BMI: Obesity group (25 kg/m2≤BMI<30 kg/m2) and severe-obesity group (BMI≥30 kg/m2).
-
Results
Of the total 266 patients, 210 patients were categorized as the obesity group and 16 as the severe-obesity group. Postoperative complications were comparable between the two groups. The BMI decreased by more than 10% at 12 months after gastrectomy, and remained at a similar level during the 4 year-follow-up period in both groups. At 4 years after surgery, the nutritional biomarkers were nearly comparable to preoperative levels in each group. The severeobesity group were associated with poor overall survival rate (P=0.011).
-
Conclusion
BMI decreased by more than 10% after gastrectomy in obese elderly, but nutritional biomarker levels were comparable before and after gastrectomy. The overall survival rate was lower in severely obese elderly than in obese elderly. Therefore, it is considered that severely obese elderly gastric cancer patients should be monitored, and vigorous efforts are needed to reduce high-BMI.
-
Keywords: Stomach neoplasms; Obesity; Aged
INTRODUCTION
Obesity rates have been increasing with social and economic developments, and the prevalence of obesity in Korean adults is reported to be 33.8% according to data from National Health Statistics 2019 [
1]. Obesity is a risk factor for negative health outcomes, such as social isolation, depression, and premature deaths as well as various diseases such as cancer, diabetes, hypertension, dislipidemia, and deep vein thrombosis [
2,
3]. Recently, as life expectancy increases, the proportion of the elderly population has increased [
1]. In addition, the prevalence of obesity in elderly people aged 65 or older is reported to be 37% or higher, and has remained at a similar level during the past decade [
1]. In particular, elderly people are a vulnerable population that shows a decrease in lean body mass and changes in body composition including the increase of body fat mass due to aging. Also, they have an increased prevalence of diseases due to multiple chronic conditions, so national health management efforts for health management and promotion in obese older people are required [
4].
In Korea, as bariatric surgery has been covered by the National Health Insurance since 2019, the necessity and usefulness of active treatment of obesity have been recognized [
5]. This bariatric surgery is known as the best treatment method in terms of long-term weight loss and the improvement of comorbidities in severely obese patients [
6]. The most commonly performed types of bariatric surgery are sleeve gastrectomy and Roux-en-Y gastric bypass [
6]. Sleeve gastrectomy is a surgical procedure to restrict food intake, and this surgery removes part of the stomach, leaving a portion of the stomach in a shape of a sleeve or banana. After sleeve gastrectomy is performed, the stomach cannot be stretched freely since the gastric fundus and greater curvature have been removed, so the patient can only consume a limited volume of food. Roux-en-Y gastric bypass is a surgery in which the stomach is resected to create a small gastric pouch and the small intestine is pulled up and connected to the gastric pouch. This surgery causes food to bypass most of the stomach, the duodenum, and the proximal portion of small intestine and move directly into the small intestine connected to the gastric pouch, so it has the advantage that it can aim at achieving the effect of limiting nutrient intake and absorption simultaneously.
In the treatment of obese gastric cancer patients, it may be considered to perform the treatment of obesity concurrently along with cancer treatment unlike performing bariatric surgery in patients without any gastric diseases. The incidence of gastric cancer is high in Asia, so 75% of the total gastric cancer patients worldwide are reported to be Asians [
7]. Especially in Korea, gastric cancer has the highest incidence rate among all cancers. The prevalence of gastric cancer per year, patients aged 60~69 and those aged 70~79 take up 28.1% and 25.4%, respectively, so elderly patients aged 60~79 account for about 50%, showing that the proportion of elderly patients is highest among newly diagnosed gastric cancer patients [
8]. In addition, according to a recent report of the prevalence of obesity in cancer survivors in 2021, the prevalence of obesity among elderly cancer survivors aged 65 or older was 26.9% [
9]. Since obesity causes or aggravates various metabolic diseases, leading to negative clinical outcomes [
2,
3], obesity management for cancer patients should be implemented concurrently with disease treatment. In performing the anasmotosis to bypass the duodenum and the upper jejunum during gastrectomy to treat gastric cancer and obesity simultaneously, onco-metabolic surgery to increase the length of the bypassed small intestine than in conventional surgery by using the principles of bariatric surgery has recently been attempted [
10-
12]. However, this surgical procedure can be applied to patients who are likely to achieve long-term survival by complete surgical resection, but the actual benefits of performing bariatric surgery concurrently in the surgical treatment of obese elderly patients with gastric cancer have not yet been sufficient. In particular, elderly patients have a higher incidence rate of postoperative complications than younger patients, and they have a reduced ability to recover from negative clinical results after surgery, so there is a need to establish the basis for active treatment interventions such as onco-metabolic surgery by examining the clinical course of elderly gastric cancer patients in various situations.
Therefore, this study attempted to investigate changes in body mass index (BMI) and nutritional biomarkers after radical gastrectomy for a long follow-up period and evaluate clinical outcomes in obese elderly patients with gastric cancer.
MATERIALS AND METHODS
1. Study design
This study was a retrospective study to investigate long-term changes in BMI and the clinical outcomes after radical gastrectomy in obese patients aged 65 or older who were diagnosed with gastric cancer at Seoul National University Hospital.
2. Subjects
Out of 1,584 patients who were diagnosed with gastric cancer and underwent radical gastrectomy at Seoul National University Hospital from January 1, 2014 to December 31, 2015, 610 patients were enrolled in this study. Out of the 610 patients, a total of 226 patients for whom it was possible to obtain preoperative clinical data and perform periodic follow-up evaluations at 1, 2, 3, and 4 years after surgery were finally included in this study by excluding the patients with the exclusion criteria. The exclusion criteria were as follows: American Society of Anesthesiologists (ASA) classification grade 3 or above (a severe systemic disease), presence of concurrent cancer other than gastric cancer, presence of advanced or metastatic gastric cancer treated by palliative surgery, a previous history of abdominal surgery (e.g. gastric, small bowel, or colon resection) which may affect nutritional status, and a previous history of endoscopic submucosal dissection to treat early-stage gastric cancer.
Regarding the classification of subjects into age groups, elderly patients aged 65 to 74 were defined as the younger old and those aged 75 or older as the older old. As to classification according to the degree of obesity, subjects were also divided into the obesity group (25 kg/m
2≤BMI<30 kg/m
2) and the severe-obesity group (BMI≥30 kg/m
2) based on preoperative BMI by using the reference of BMI≥25 kg/m
2 presented by WHO Asia-Pacific BMI classification and the Korean Society for the Study of Obesity [
13].
After obtaining approval from the Institutional Review Board of Seoul National University Hospital (IRB No.:1808-070-965), data collection was carried out using the computerized medical record program as follows. Informed consent was waived because of the retrospective study design.
With respect to demographic characteristics of the subjects, data on age, gender, and comorbidity were collected. Regarding disease characteristics, diagnosis, cancer stage (AJCC 8th), hospital admission date, operation date, discharge date, length of hospital stay, surgical method, type of adjuvant chemotherapy, type of hospital discharge, ASA grade, occurrence of complications, types of complications, Clavien-Dindo classification, presence of postoperative recurrence, recurrence date, recurrence site, death status, and death date were examined. Regarding nutritional characteristics, the subjects’ height, body weight at each follow-up timepoint, body weight measurement date, and BMI were investigated. As to nutritional biochemical markers, hemoglobin, albumin, and total protein levels at each follow-up timepoint were examined.
4. Data analysis
The statistical analysis of collected data was carried out using the IBM SPSS/WIN version 23.0 (IBM Co., Armonk, NY, USA). The characteristics of the subjects were analyzed using frequency, percentage, mean, and standard deviation. The characteristics of the subjects according to age were analyzed by performing the one-way ANOVA. In addition, differences in BMI and clinical parameters according to categorized characteristics of the subjects were analyzed using the chi-squared test. For variables with a non-normal distribution, significance was verified using the Mann-Whitney test and Kruskal-Wallis test. The level of significance was defined as P<0.05. The Kaplan-Meier estimate was used to analyze the mortality rate according to age and the degree of obesity.
RESULTS
1. Characteristics of according to the age and degree of obesity
With respect to the classification of the subjects into age groups, elderly patients aged 65 to 74 were defined as the younger old group and those aged 75 or older were categorized as the older old group. The younger old group took up 76% (n=173), and older old group made up 24% (n=53) (
Table 1). In terms of obesity, subjects were classified into the obesity group (25 kg/m
2≤BMI<30 kg/m
2) and the severe-obesity group (BMI≥30 kg/m
2) based on preoperative BMI by using BMI≥25 kg/m
2 as the reference. The obesity group and the severe-obesity group made up 92% (n=210) and 8% (n=16) of the total subjects, respectively (
Table 2).
The analysis results of differences between pre- and post-operative BMI levels in the younger old and older old groups are shown in
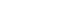
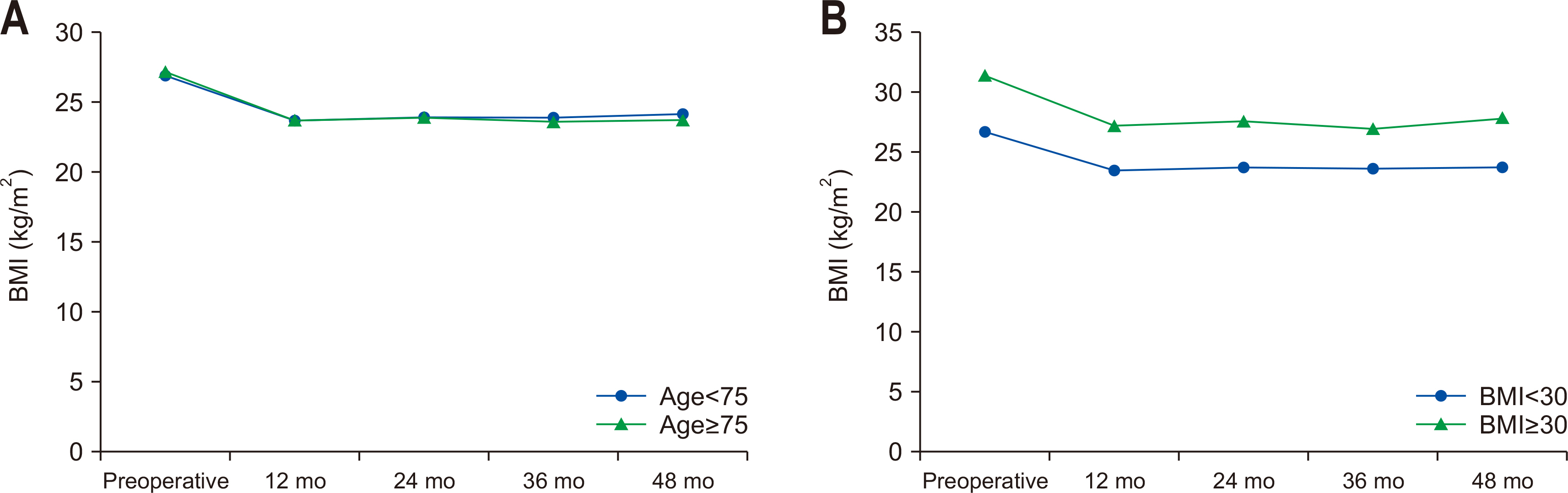
Fig. 1A. In both groups, BMI decreased by about 10% at 12 months after surgery compared to preoperative BMI, and BMI remained at a similar level during the 4-year follow-up period. There were no significant differences between groups.
Based on the preoperative BMI, subjects were classified into the obesity and severe-obesity groups, and analysis results of changes in BMI after surgery compared to preoperative BMI are shown in
Fig. 1B. The preoperative mean BMI was 26.7 kg/m
2 in the obesity group and 31.4 kg/m
2 in the severe-obesity group. In the severe-obesity group, the postoperative mean BMI remained as high as 27.7 kg/m
2 at four years after surgery.
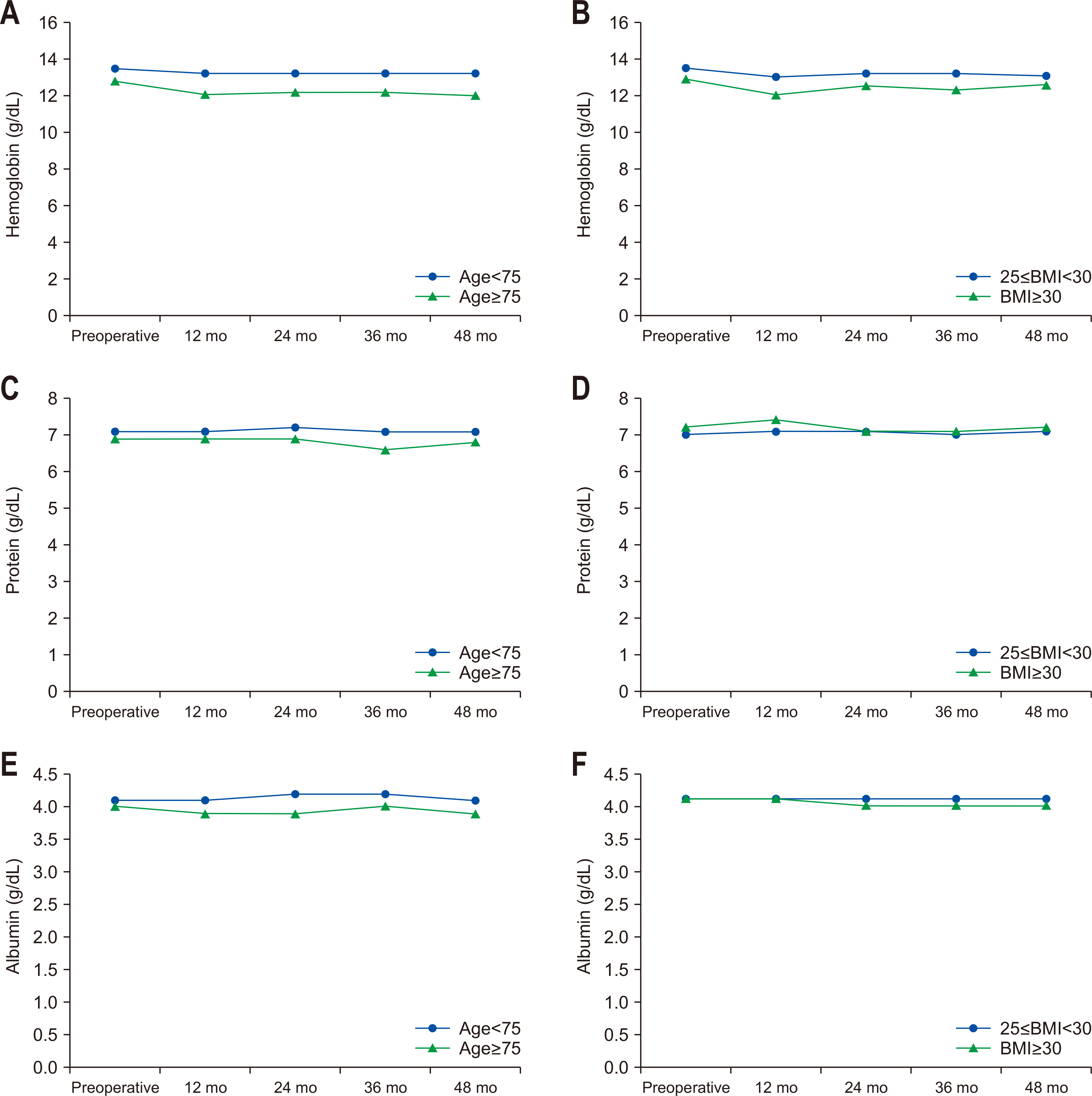
The analysis results of hemoglobin, albumin, and total protein levels according to age and degree of obesity in the subjects are as follows (
Fig. 2). In hemoglobin and albumin levels according to age, the preoperative hemoglobin levels of the younger old and older old groups were 13.6±1.8 g/dL and 12.9±1.6 g/dL (P=0.008), respectively, and the preoperative serum albumin levels of the two groups were 4.1±0.4 g/dL and 4.0±10.4 g/dL (P=0.060), respectively. Compared to the younger old group, the older old group showed a decrease in hemoglobin and albumin levels at 12 months after surgery (younger old 13.2±1.6 g/dL vs. older old 11.9±1.6 g/dL, P<0.001), but hemoglobin and albumin levels remained at a similar level during the 4-year follow-up period. Regarding the total protein level, the preoperative total protein level was lower in the older old group than the younger old group (younger old 7.1±0.6 g/dL vs. older old 6.9±0.7 g/dL, P=0.004), and the total protein level remained at a similar level during the 4-year follow-up period (younger old 7.1±0.4 g/dL vs. older old 6.8±0.4 g/dL, P=0.002).
There were no significant differences between the obesity group and severe-obesity group, and hemoglobin, albumin, and total protein levels remained similar to the preoperative levels even at four years after surgery.
5. Overall survival according to age and degree of obesity
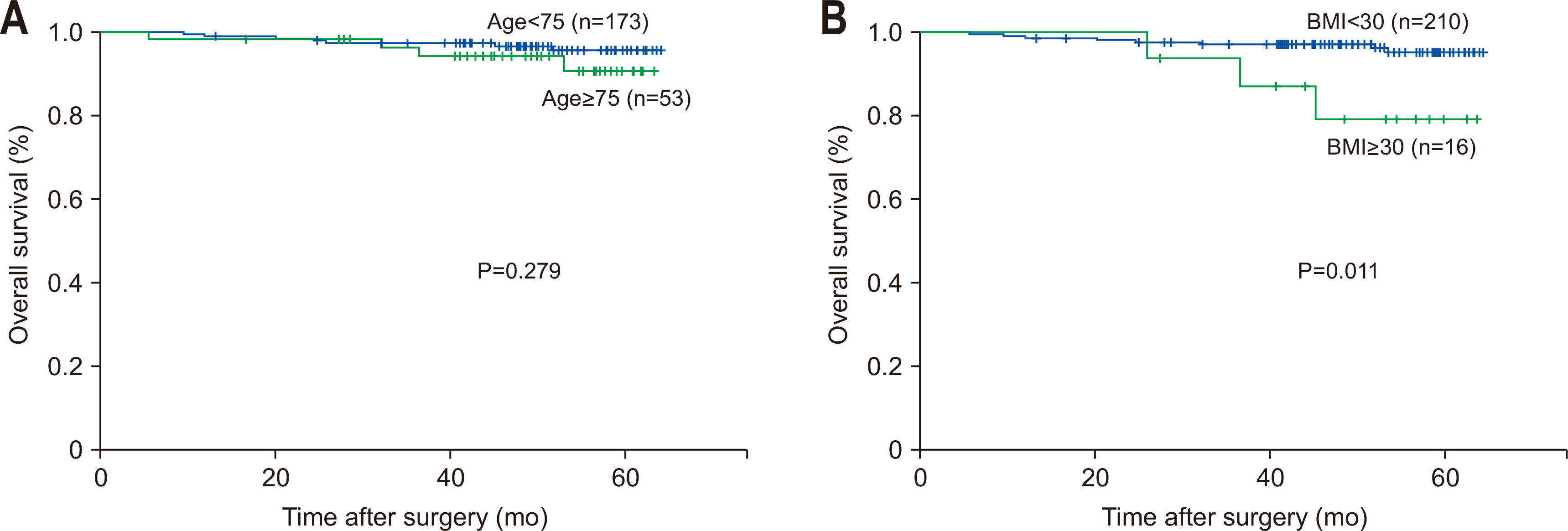
The analysis results of overall survival according to age and the degree of obesity are as follows (
Fig. 3). Regarding overall survival according to age, there were no significant differences in overall survival between the younger old and older old groups (95.9% vs. 89.9%, P=0.279). As for overall survival according to the degree of obesity, there was a significant difference in overall survival between the obesity and severe-obesity groups (95.5% vs. 80.0%, P=0.011).
DISCUSSION
With respect to the health management of cancer patients who are likely to achieve long-term survival by surgical treatment, obesity is likely to cause and aggravate various metabolic diseases, leading to negative clinical outcomes [
2,
3]. Thus, onco-metabolic surgery has recently been attempted to treat gastric cancer and obesity simultaneously in obese gastric cancer patients [
10-
12]. However, actual benefits of concurrent bariatric surgery in the surgical treatment of obese elderly patients with gastric cancer have not been insufficient. Therefore, this study aimed to investigate long-term changes in BMI and nutritional biomarkers after radical gastrectomy in obese elderly patients with gastric cancer in order to provide basic data for clinical decision making related to gastric cancer treatment in obese elderly patients.
In this discussion, obese elderly patients with gastric cancer are classified according to age and degree of obesity, and differences in clinical characteristics between groups are comparatively examined.
In terms of age, elderly patients aged below 75 were categorized as the younger old group and those aged 75 or older as the older old group. In both age groups, the mean BMI was about 27.0 kg/m
2 before surgery and about 23.7 kg/m
2 at 12 months after surgery. The mean BMI was decreased by about 13% at 12 months after surgery, compared to preoperative BMI, and it remained at a similar level during the 4-year follow-up period. There were no significant differences in the mean BMI between the two age groups at each follow-up timepoint. These results are similar to the findings of previous studies reporting the tendency that preoperative body weight decreased by an average of 5%~10% at 12 months after radical gastrectomy and the level of body weight was maintained during a long period [
14,
15]. Thus, the results of the present study confirmed that age of elderly patients does not have a significant effect on differences between pre- and post-operative BMI in obese elderly gastric cancer patients.
The clinical characteristics according to age revealed that there were no differences in the incidence of complications and the survival rate according to age. Although it has been found that age is generally one of the factors affecting the morbidity of diseases in older people, some studies reported that age was shown to have no significant effect on the incidence of complications in gastric cancer patients who underwent gastrectomy, while malnutrition was found to be a main influencing factor, and poorer nutritional status was associated with a higher incidence of complications [
16,
17]. In this study, regarding postoperative complications in obese elderly patients with gastric cancer, age was not a significant influencing factor for differences between groups. However, in both the younger old and older old groups, out of the total postoperative complications, complications with Clavian-Dindo Classification grade 2 or above took up 80% or more. In particular, the incidence of complications with Clavian-Dindo Classification grade 3 or above was slightly higher in the older old group than the younger old group, showing that obese elderly patients in the older old group are vulnerable to severe complications. Therefore, for obese elderly patients in the older old group, careful attention should be paid to health management including complications after gastrectomy.
In some nutritional biochemical markers, there were differences between the two age groups. In particular, the older old group showed significantly lower hemoglobin and albumin levels than the younger old group before surgery and until four years after surgery. A previous investigation of the long-term clinical outcomes after gastrectomy also reported that nutritional biomarkers remained at similar levels within a normal range without significant changes at 12 months after surgery [
14], and the results of the present study showed a similar tendency. Consistent with the previous research, in this study, nutritional biochemical markers in the obese elderly patients with gastric cancer remained at a similar level within a normal range during the 4-year follow-up period after surgery, but the older old group continued to show slightly lower nutritional biomarker levels and were more vulnerable in terms of the degree of recovery than the younger old group. These results indicate that there is a need to carefully monitor changes in nutritional biochemical markers in obese elderly patients aged 75 or older.
Elderly gastric cancer patients were divided into the obesity group (25 kg/m
2≤BMI<30 kg/m
2) and the severe-obesity group (BMI≥30 kg/m
2) according to preoperative BMI, and comparative analysis of changes in BMI after surgery in the two groups found that both groups showed a decrease in BMI by about 12% at 12 months after surgery. In addition, in the severe-obesity group, mean BMI remained at about 27~28 kg/m
2 for a long period after surgery. A BMI-loss trajectory model study to examine long-term BMI changes in gastric cancer patients, in the obesity group (preoperative mean BMI≥26 kg/m
2) reported that the BMI level was decreased by approximately an average of 20% after surgery [
18]. In comparison, the obesity group in this study showed a relatively smaller decrease in BMI. On the other hand, elderly gastric cancer patients in the severe-obesity group continued to have BMI levels in the obese range during the follow-up period despite a decrease in BMI by 10% or more after surgery. These results suggest that there is a need to consider implementing intensive obesity management for at least one year after surgery. Therefore, in obese elderly patients with gastric cancer, clinical management strategies differentiated from those for nonobese elderly patients are required to maintain adequate body weight by applying a postoperative management method after surgery. In particular, in elderly patients, a dietetic intervention for weight loss after gastrectomy is highly likely to cause nutritional imbalance, which may lead to an increase in the morbidity rates of various diseases, and aggravate disease in elderly patients with multiple chronic conditions [
19,
20]. Therefore, to prevent the reduction of bone mass and nutrient deficiencies including mineral deficiencies which may occur during long-term dietetic therapy, multidisciplinary efforts should also be made concurrently along with disease treatment. In addition, personalized interventions are needed that take into account the level of physical activity according to the age.
It has been found that there are no significant differences between groups in the incidence of complications and nutritional biomarker levels according to the degree of obesity. Obesity is known to be associated with the increased preoperative risk of cardiovascular and pulmonary comorbidities, thereby increasing the risk of perioperative and postoperative complications [
21]. In addition, although it has been reported that obesity is associated with the increased incidence of complications including postoperative wound infections and has a negative effect on the survival rate in gastric cancer patients [
22-
24]. However, some studies reported that obesity did not have any effects on postoperative complications and the survival rate [
25,
26], suggesting that the impact of obesity may vary depending on the circumstances of patients. In this study, the analysis of overall survival according to the degree of obesity found that the overall survival rate was significantly lower in the severe-obesity group with a BMI≥30 kg/m
2. Obesity is one of the factors affecting the prevalence of various diseases and is associated especially with cancer mortality [
3,
22-
25,
27]. However, there has been a lack of studies on the association between obesity and cancer in Korea. Some previous studies reported the occurrence of gastric cancer in the remnant blind stomach in patients who underwent bariatric surgery [
28,
29]. Another research showed that patients with early-stage gastric cancer had a significantly higher BMI than those with advanced gastric cancer [
30]. Although there still have been few studies on the association between gastric cancer and obesity in vulnerable populations such as elderly people. Therefore, further studies on the association between gastric cancer and obesity are needed in future studies.
As discussed above, in severely obese elderly patients with gastric cancer, positive clinical outcomes can be expected if continuous obesity management is implemented after surgery.
Since this research is a retrospective study, one of the limitations of the present study is that all the factors potentially influencing clinical outcomes in obese elderly gastric cancer patients, such as the severity of comorbidity, the presence of medical management of diabetes, and socioeconomic status, were not taken into account. However, this study attempted to provide basic data for clinical decision making for gastric cancer treatment in obese elderly patients by classifying obese elderly gastric cancer patients according to age and preoperative BMI and analyzing changes in BMI and biochemical markers.
CONCLUSION
In obese elderly gastric cancer patients, BMI was decreased by more than 10% after gastrectomy, but the severe-obesity group continued to have the BMI level in the obese range even after surgery. Although the levels of biochemical markers were slightly higher in the younger old group than the older old group, the biomarker levels of the two groups were comparable within a normal range before and after surgery. The severe-obesity group showed poorer overall survival than the obesity group. These study results indicate that more careful monitoring is required after gastrectomy for severely obese elderly gastric cancer patients, and active efforts for obesity management should be made even after surgery.
AUTHOR CONTRIBUTIONS
Conceptualization: EK, HJL. Data curation: EK, HJL. Formal analysis: EK, JHP. Methodology: EK, JHP, EMS. Project administration: HJL. Resources: SHK, DJP, HKY, HJL. Software: EK. Supervision: JHP, SHK, DJP, HKY, HJL. Visualization: EK, HJL. Writing – original draft: EK. Writing – review & editing: EK, JHP, HJL.
CONFLICTS OF INTEREST
The authors of this manuscript have no conflicts of interest to disclose.
Fig. 1(A) Change of BMI according to age group. (B) Change of BMI according to obesity group. BMI = body mass index.
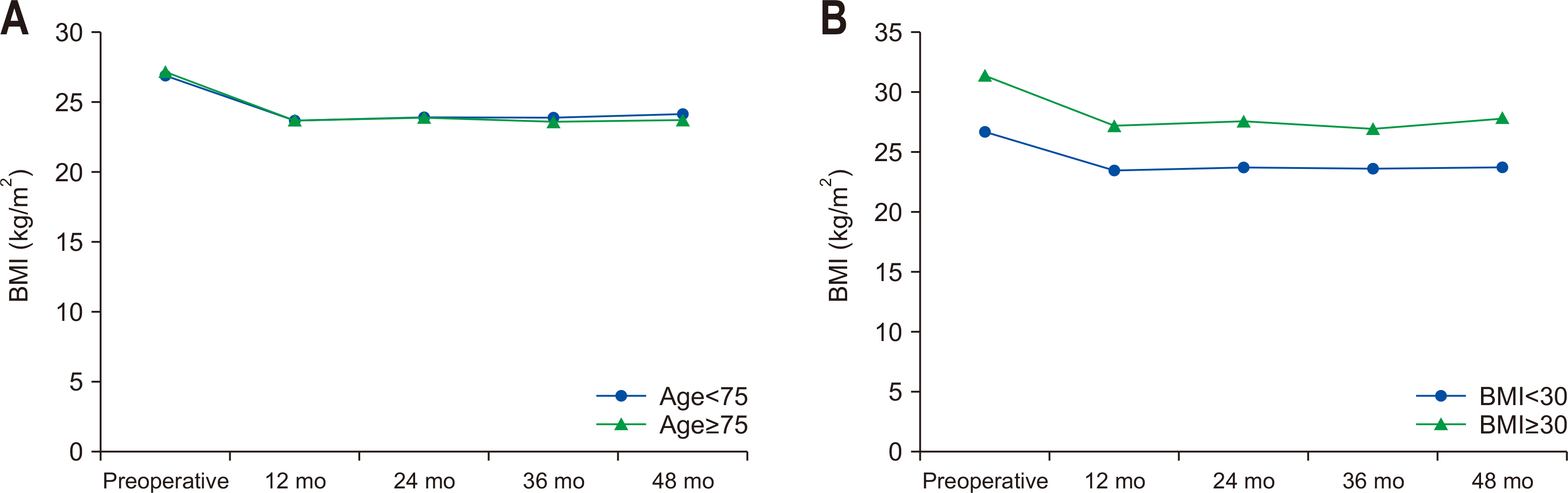
Fig. 2(A) Change of serum hemoglobin according to age. (B) Change of serum hemoglobin according to obesity group. (C) Change of serum protein according to age. (D) Change of serum protein according to obesity group. (E) Change of serum albumin according to age. (F) Change of serum albumin according to obesity group. BMI = body mass index.
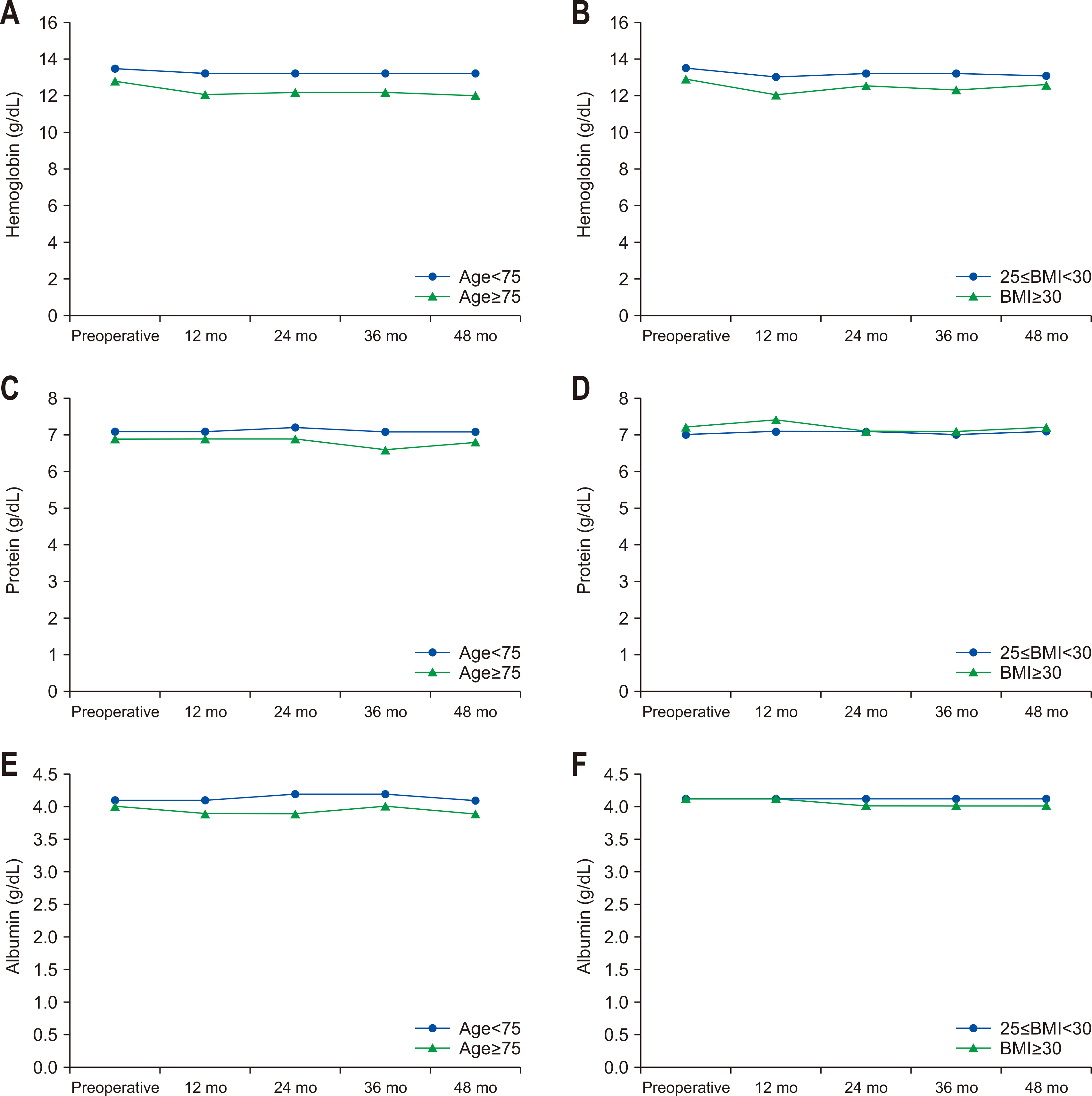
Fig. 3(A) Oveall survival in age group. (B) Overall survival in obesity group. BMI = body mass index.
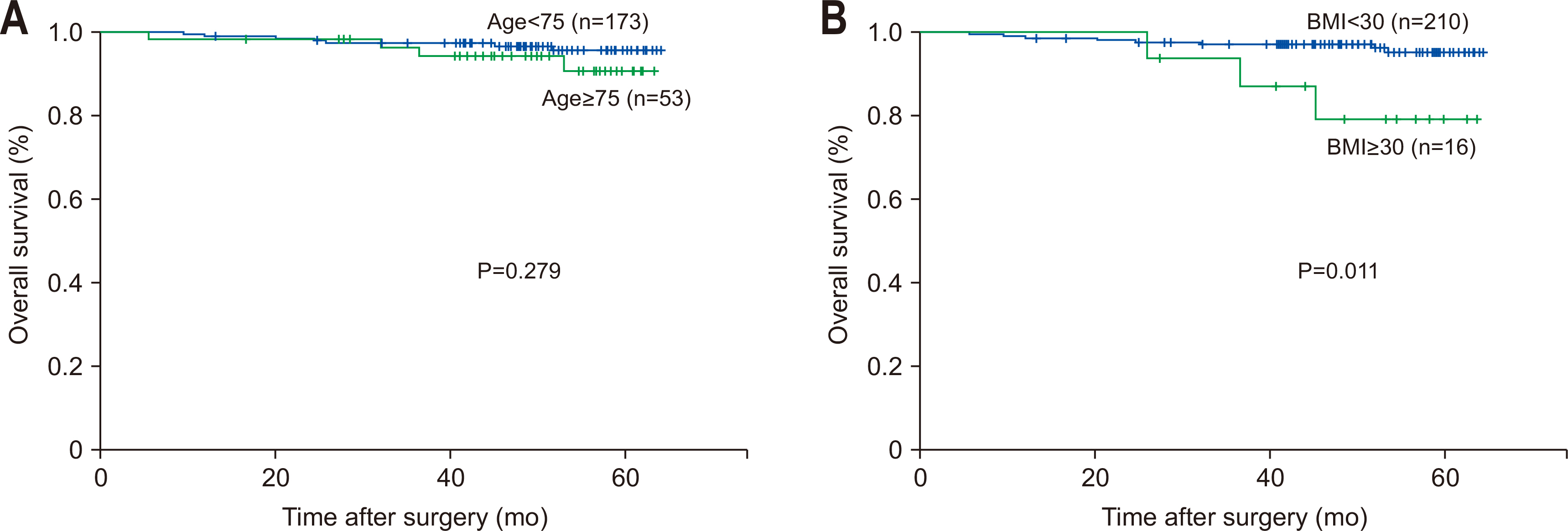
Table 1Characteristics according to age group
|
Variables |
Overall |
Age (yr) |
|
|
<75 (n=173) |
≥75 (n=53) |
P-value |
|
Age (yr) |
71.4±4.9 |
69.0±2.9 |
78.4±3.1 |
<0.001 |
|
Sex |
|
|
|
0.002 |
|
Male |
135 (59.7) |
113 (65.3) |
22 (41.5) |
|
|
Female |
91 (40.3) |
60 (34.7) |
31 (58.5) |
|
|
BMI (kg/m2) |
|
|
|
0.879 |
|
25≤BMI<30 |
210 (92.9) |
161 (93.1) |
49 (92.5) |
|
|
BMI ≥30 |
16 (7.1) |
12 (6.9) |
4 (7.5) |
|
|
Hospital stay (day) |
12.1±12.3 |
12.3±14.2 |
13.4±12.3 |
0.621 |
|
Adjuvant therapy |
|
|
|
0.240 |
|
None |
205 (90.7) |
263 (90.7) |
86 (89.6) |
|
|
TS-1 |
9 (4.0) |
5 (2.9) |
4 (7.5) |
|
|
XELOX |
6 (2.7) |
6 (3.5) |
0 (0.0) |
|
|
Others |
6 (2.7) |
5 (2.9) |
1 (1.9) |
|
|
Previous DM |
57 (10.5) |
52 (10.7) |
5 (9.1) |
0.720 |
|
Cardiovascular |
190 (35.0) |
179 (36.7) |
11 (20.0) |
0.014 |
|
No. of comorbidity |
|
|
|
0.203 |
|
0 |
41 (18.1) |
35 (16.7) |
6 (37.5) |
|
|
1 |
89 (39.4) |
85 (40.5) |
4 (25.0) |
|
|
2 |
60 (26.5) |
56 (26.7) |
4 (25.0) |
|
|
More than 3 |
36 (15.9) |
34 (16.2) |
2 (12.5) |
|
|
No. of comorbidity (mean) |
1.5±1.1 |
1.5±1.1 |
1.2±1.2 |
0.243 |
|
Operation |
|
|
|
0.719 |
|
DG |
64 (28.3) |
49 (28.3) |
15 (28.3) |
|
|
TG |
77 (34.1) |
57 (32.9) |
20 (37.7) |
|
|
PG |
69 (30.5) |
53 (30.6) |
16 (30.2) |
|
|
PPG |
16 (7.1) |
14 (8.1) |
2 (3.8) |
|
|
Operation method |
|
|
|
0.203 |
|
Open |
148 (65.8) |
111 (64.5) |
37 (69.8) |
|
|
Lapa-assisted |
43 (19.1) |
37 (21.5) |
6 (11.3) |
|
|
Total-laparoscopy |
3 (1.3) |
3 (1.7) |
0 (0.0) |
|
|
Robot |
31 (13.8) |
21 (12.2) |
10 (18.9) |
|
|
Postop. Complication |
62 (27.4) |
46 (26.6) |
16 (30.2) |
0.607 |
|
Clavien-Dindo grade |
|
|
|
0.356 |
|
Grade I |
9 (14.5) |
6 (13.0) |
3 (18.8) |
|
|
Grade II |
25 (40.3) |
17 (37.0) |
8 (50.0) |
|
|
Grade IIIa |
22 (35.5) |
19 (41.3) |
3 (18.8) |
|
|
Grade IIIb |
4 (6.5) |
2 (4.3) |
2 (12.5) |
|
|
Grade IV |
2 (3.2) |
2 (4.3) |
0 (0.0) |
|
|
Cancer stage |
|
|
|
0.700 |
|
0 |
4 (1.8) |
4 (2.3) |
0 (0.1) |
|
|
IA |
119 (53.1) |
95 (55.6) |
24 (45.3) |
|
|
IB |
33 (14.7) |
23 (13.5) |
10 (18.9) |
|
|
IIA |
16 (7.1) |
11 (6.4) |
5 (9.4) |
|
|
IIB |
18 (8.0) |
14 (8.2) |
4 (7.5) |
|
|
IIIA |
17 (7.6) |
11 (6.4) |
6 (11.3) |
|
|
IIIB |
9 (4.0) |
7 (4.1) |
2 (3.8) |
|
Table 2Characteristics according to obesity group
|
Variables |
Overall |
Preoperative BMI (kg/m2) |
|
|
25≤BMI<30 (n=210) |
BMI ≥30 (n=16) |
P-value |
|
Age (yr) |
71.4±4.9 |
71.2±5.1 |
71.5±3.9 |
0.548a
|
|
Sex |
173 (76.5) |
161 (76.7) |
12 (75.0) |
0.879 |
|
Male |
53 (23.5) |
49 (23.3) |
4 (25) |
|
|
Female |
|
|
|
|
|
BMI (kg/m2) |
135 (59.7) |
130 (61.9) |
5 (31.3) |
0.016 |
|
25≤BMI<30 |
91 (40.3) |
80 (38.1) |
11 (68.8) |
<0.001a
|
|
BMI ≥30 |
27.0±1.8 |
26.7±1.3 |
31.4±1.7 |
|
|
Hospital stay (days) |
12.6±13.8 |
12.4±13.9 |
14.3±11.0 |
0.330 |
|
Adjuvant Tx |
|
|
|
0.674 |
|
None |
205 (90.7) |
191 (91.0) |
14 (87.5) |
|
|
TS-1 |
9 (4.0) |
8 (3.8) |
1 (6.3) |
|
|
XELOX |
6 (2.7) |
6 (2.9) |
0 (0.0) |
|
|
Others |
6 (2.7) |
5 (2.4) |
1 (6.3) |
|
|
Previous DM |
49 (21.7) |
44 (21.0) |
5 (31.3) |
0.335 |
|
Cardiovascular |
160 (70.8) |
149 (71.0) |
11 (68.8) |
0.852 |
|
No. of comorbidity |
|
|
|
0.203 |
|
0 |
41 (18.1) |
35 (16.7) |
6 (37.5) |
|
|
1 |
89 (39.4) |
85 (40.5) |
4 (25.0) |
|
|
2 |
60 (26.5) |
56 (26.7) |
4 (25.0) |
|
|
More than 3 |
36 (15.9) |
34 (16.2) |
2 (12.5) |
|
|
No of comorbidity (mean) |
1.5±1.1 |
1.5±1.1 |
1.2±1.2 |
0.243 |
|
Operation |
|
|
|
0.276 |
|
DG |
64 (28.3) |
58 (27.6) |
6 (37.5) |
|
|
TG |
77 (34.1) |
75 (35.7) |
2 (12.5) |
|
|
PG |
69 (30.5) |
62 (29.5) |
7 (43.7) |
|
|
PPG |
16 (7.1) |
15 (7.15) |
1 (6.3) |
|
|
Operation method |
|
|
|
0.001 |
|
Open |
148 (65.8) |
137 (65.6) |
11 (68.8) |
|
|
Lapa-assisted |
43 (19.1) |
41 (19.6) |
2 (12.5) |
|
|
Total-laparoscopy |
3 (1.3) |
1 (0.5) |
2 (12.5) |
|
|
Robot |
31 (13.8) |
30 (14.4) |
1 (6.3) |
|
|
Postop. Complication |
62 (27.4) |
56 (26.7) |
6 (37.5) |
0.349 |
|
Clavien-Dindo grade |
|
|
|
0.003 |
|
Grade I |
9 (14.5) |
5 (8.9) |
4 (66.7) |
|
|
Grade II |
25 (40.3) |
25 (44.6) |
0 (0.0) |
|
|
Grade IIIa |
22 (35.5) |
20 (35.7) |
2 (33.3) |
|
|
Grade IIIb |
4 (6.5) |
4 (7.1) |
0 (0.0) |
|
|
Grade IV |
2 (3.2) |
2 (3.6) |
0 (0.0) |
|
|
Cancer stage |
|
|
|
0.957 |
|
0 |
4 (1.8) |
4 (1.9) |
0 (0.0) |
|
|
IA |
119 (53.1) |
111 (53.4) |
8 (50.0) |
|
|
IB |
33 (14.7) |
30 (14.4) |
3 (18.8) |
|
|
IIA |
16 (7.1) |
15 (7.2) |
1 (6.3) |
|
|
IIB |
18 (8.0) |
16 (7.7) |
2 (12.5) |
|
|
IIIA |
17 (7.6) |
16 (7.7) |
1 (6.3) |
|
|
IIIB |
9 (4.0) |
9 (4.3) |
0 (0.0) |
|
|
IIIC |
8 (3.6) |
7 (3.4) |
1 (6.3) |
|
References
- 1. Organisation for Economic Cooperation and Development. 2019. Health at a glance 2019 [Internet]. OECD Publishing; Paris: Available from: http://www.oecd.org/els/health-systems/health-at-a-glance. [cited 2019 Feb 28
- 2. Bray GA. Complications of obesity. Ann Intern Med 1985;103:1052-62. ArticlePubMedPDF
- 3. Calle EE, Kaaks R. Overweight, obesity and cancer: epidemiological evidence and proposed mechanisms. Nat Rev Cancer 2004;4:579-91. ArticlePubMedPDF
- 4. Stenholm S, Harris TB, Rantanen T, Visser M, Kritchevsky SB, Ferrucci L. Sarcopenic obesity: definition, cause and consequences. Curr Opin Clin Nutr Metab Care 2008;11:693-700. ArticlePubMedPMC
- 5. Ministry of Health and Welfare. 2018. Comprehensive measures for national obesity management [Internet]. Ministry of Health and Welfare; Sejong: Available from: http://www.mohw.go.kr/react/al/sal0301vw.jsp?PAR_MENU_ID=04&MENU_ID=0403&CONT_SEQ=345515&page=1. [cited 2019 Mar 1
- 6. Angrisani L, Santonicola A, Iovino P, Vitiello A, Zundel N, Buchwald H, et al. Bariatric surgery and endoluminal procedures: IFSO worldwide survey 2014. Obes Surg 2017;27:2279-89. ArticlePubMedPMCPDF
- 7. Korea Central Cancer Registry, National Cancer Center. 2017. Annual report of cancer statistics in Korea in 2015 [Internet]. Ministry of Health and Welfare; Sejong: Available from: https://ncc.re.kr/cancerStatsView.ncc?bbsnum=438&searchKey=total&searchValue=&pageNum=1. [cited 2018 Apr 24
- 8. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018;68:394-424. ArticlePubMedPDF
- 9. National Cancer Center. 2021. Obesity prevalence in cancer survivor [Internet]. National Cancer Center; Goyang: Available from: https://www.cancerdata.kr/surveillance/data;jsessionid=E193331CCE90F2962B3B9C0737320FFF?menuId=49#n. [cited 2021 Mar 5
- 10. Lee CM, Kim JH. Surgical treatment of morbid obesity. Korean J Helicobacter Up Gastrointest Res 2017;17:72-8. ArticlePDF
- 11. Kim WS, Kim JW, Ahn CW, Choi SH. Resolution of type 2 diabetes after gastrectomy for gastric cancer with long limb Roux-en Y reconstruction: a prospective pilot study. J Korean Surg Soc 2013;84:88-93. ArticlePubMedPMC
- 12. Kwon Y, Abdemur A, Lo Menzo E, Park S, Szomstein S, Rosenthal RJ. The foregut theory as a possible mechanism of action for the remission of type 2 diabetes in low body mass index patients undergoing subtotal gastrectomy for gastric cancer. Surg Obes Relat Dis 2014;10:235-42. ArticlePubMed
- 13. WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 2004;363:157-63. ArticlePubMed
- 14. Shin BS, Kim DY, Nam SH, Yook JH, Oh ST, Kim BS. Correlation of the body mass index with the rates of postoperative wound complications in gastric cancer patients. J Korean Gastric Cancer Assoc 2007;7:242-7. ArticlePDF
- 15. Na JR, Suh YS, Kong SH, Lim JH, Ju DL, Yang HK, et al. A prospective observational study evaluating the change of nutritional status and the incidence of dumping syndrome after gastrectomy. J Clin Nutr 2014;6:59-70. Article
- 16. Kim Y, Kim WG, Lee HJ, Park MS, Lee YH, Kong SH, Yang HK. Comparison of the impact of malnutrition by nutritional assessment and screening tools on operative morbidity after gastric cancer surgery. J Korean Soc Parenter Enter Nutr 2011;4:7-15. Article
- 17. Shin DG, Seong SH, Kim WS, Kim DS. The effect of old age on the development of postoperative complication in surgery for gastric carcinoma. J Korean Surg Soc 2005;69:455-8.
- 18. Park JH, Kim E, Seol EM, Kong SH, Park DJ, Yang HK, et al. Prediction model for screening patients at risk of malnutrition after gastric cancer surgery. Ann Surg Oncol 2021;28:4471-81. ArticlePubMedPDF
- 19. Ministry of Health and Welfare. 2020. The status of welfare facilities for the elderly 2020 [Internet]. Ministry of Health and Welfare; Sejong: Available from:http://www.mohw.go.kr. [cited 2020 Jul 30
- 20. Han G. Evaluation of chronic disease and nutritional intake by obesity of Korean elderly - data from Korea National Health and Nutrition Examination Survey 2016~2018 -. Korean J Food Nutr 2020;33:428-39.
- 21. Lee H. Anesthetic management of the bariatric surgery. J Korean Med Assoc 2012;55:996-1002. Article
- 22. Inagawa S, Adachi S, Oda T, Kawamoto T, Koike N, Fukao K. Effect of fat volume on postoperative complications and survival rate after D2 dissection for gastric cancer. Gastric Cancer 2000;3:141-4. ArticlePubMedPDF
- 23. Moriwaki Y, Kunisaki C, Kobayashi S, Harada H, Imai S, Kasaoka C. Does body mass index (BMI) influence morbidity and long-term survival in gastric cancer patients after gastrectomy? Hepatogastroenterology 2003;50:284-8. PubMed
- 24. Shin BS, Kim DY, Nam SH, Yook JH, Oh ST, Kim BS. Correlation of the body mass index with the rates of postoperative wound complications in gastric cancer patients. J Korean Gastric Cancer Assoc 2007;7:242-7. ArticlePDF
- 25. Gretschel S, Christoph F, Bembenek A, Estevez-Schwarz L, Schneider U, Schlag PM. Body mass index does not affect systematic D2 lymph node dissection and postoperative morbidity in gastric cancer patients. Ann Surg Oncol 2003;10:363-8. ArticlePubMedPDF
- 26. Barry JD, Blackshaw GR, Edwards P, Lewis WG, Murphy P, Hodzovic I, et al. Western body mass indices need not compromise outcomes after modified D2 gastrectomy for carcinoma. Gastric Cancer 2003;6:80-5. ArticlePubMedPDF
- 27. Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med 2003;348:1625-38. ArticlePubMed
- 28. Escalona A, Guzmán S, Ibáñez L, Meneses L, Huete A, Solar A. Gastric cancer after Roux-en-Y gastric bypass. Obes Surg 2005;15:423-7. ArticlePubMedPDF
- 29. Corsini DA, Simoneti CA, Moreira G, Lima SE Jr, Garrido AB. Cancer in the excluded stomach 4 years after gastric bypass. Obes Surg 2006;16:932-4. ArticlePubMed
- 30. Kim YS, Park HA, Kim BS, Yook JH, Lee MS. Efficacy of screening for gastric cancer in a Korean adult population: a case-control study. J Korean Med Sci 2000;15:510-5. ArticlePubMedPMC

 , Ji-Hyeon Park, M.D., Ph.D.2
, Ji-Hyeon Park, M.D., Ph.D.2 , Eun-Mi Seol, R.N., Ph.D.1,3
, Eun-Mi Seol, R.N., Ph.D.1,3 , Seong-Ho Kong, M.D., Ph.D.2
, Seong-Ho Kong, M.D., Ph.D.2 , Do Joong Park, M.D., Ph.D.2
, Do Joong Park, M.D., Ph.D.2 , Han-Kwang Yang, M.D., Ph.D.2
, Han-Kwang Yang, M.D., Ph.D.2 , Hyuk-Joon Lee, M.D., Ph.D.2
, Hyuk-Joon Lee, M.D., Ph.D.2





 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN Cite
Cite