Scopus, KCI, KoreaMed

Articles
- Page Path
- HOME > Ann Clin Nutr Metab > Volume 18(1); 2026 > Article
- Original Article Association of soy oil-based lipid injectable emulsion with early body weight loss after minimally invasive esophagectomy in Japan: a retrospective cohort study
-
Tomonori Narita1,2
 , Kazuhiko Fukatsu2,3
, Kazuhiko Fukatsu2,3 , Kenichi Kono3
, Kenichi Kono3 , Satoshi Murakoshi3,4
, Satoshi Murakoshi3,4 , Reo Inoue3
, Reo Inoue3 , Midori Noguchi3
, Midori Noguchi3 , Nana Matsumoto2
, Nana Matsumoto2 , Shoh Yajima1
, Shoh Yajima1 , Koichi Yagi1
, Koichi Yagi1 , Yoshifumi Baba1
, Yoshifumi Baba1
-
Annals of Clinical Nutrition and Metabolism 2026;18(1):34-43.
DOI: https://doi.org/10.15747/ACNM.25.0030
Published online: March 30, 2026
1Department of Gastrointestinal Surgery, Graduate School of Medicine, The University of Tokyo, Tokyo, Japan
2Operating Room Management and Surgical Metabolism, Graduate School of Medicine, The University of Tokyo, Tokyo, Japan
3Surgical Center, The University of Tokyo Hospital, Tokyo, Japan
4Faculty of Health and Social Work, School of Nutrition and Dietetics, Kanagawa University of Human Services, Kanagawa, Japan
- Corresponding author: Tomonori Narita, e-mail: tomnrt@outlook.jp
© 2026 The Korean Society of Surgical Metabolism and Nutrition · The Korean Society for Parenteral and Enteral Nutrition · Asian Society of Surgical Metabolism and Nutrition
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 681 Views
- 13 Download
Abstract
-
Purpose Postoperative body weight loss (PBWL) is associated with poor long-term outcomes following esophagectomy for esophageal cancer, underscoring the critical importance of perioperative nutritional management. Although minimally invasive procedures, such as robot-assisted radical transmediastinal esophagectomy (RA-TME), have become increasingly prevalent, perioperative nutritional strategies have received comparatively limited attention. This study evaluated the impact of soy oil-based injectable lipid emulsion (SO-ILE) on PBWL in patients undergoing RA-TME.
-
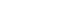
Methods We retrospectively analyzed 155 patients who underwent RA-TME for esophageal or esophagogastric junction cancer at our hospital between 2011 and 2022. Patients were divided into two groups: the lipid (+) group (n=33), which received SO-ILE between postoperative days 1 and 6, and the lipid (–) group (n=122), which did not receive SO-ILE. Oral or enteral nutrition was withheld until postoperative day 6. Nutrient intake, postoperative outcomes, and nutritional status were compared between the two groups.
-
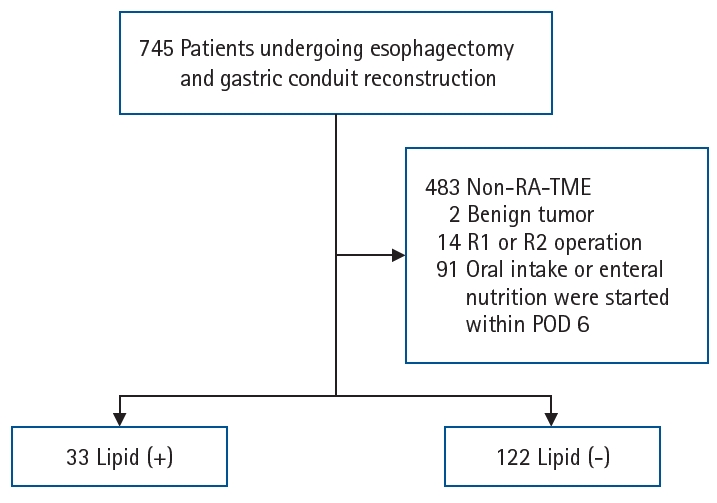
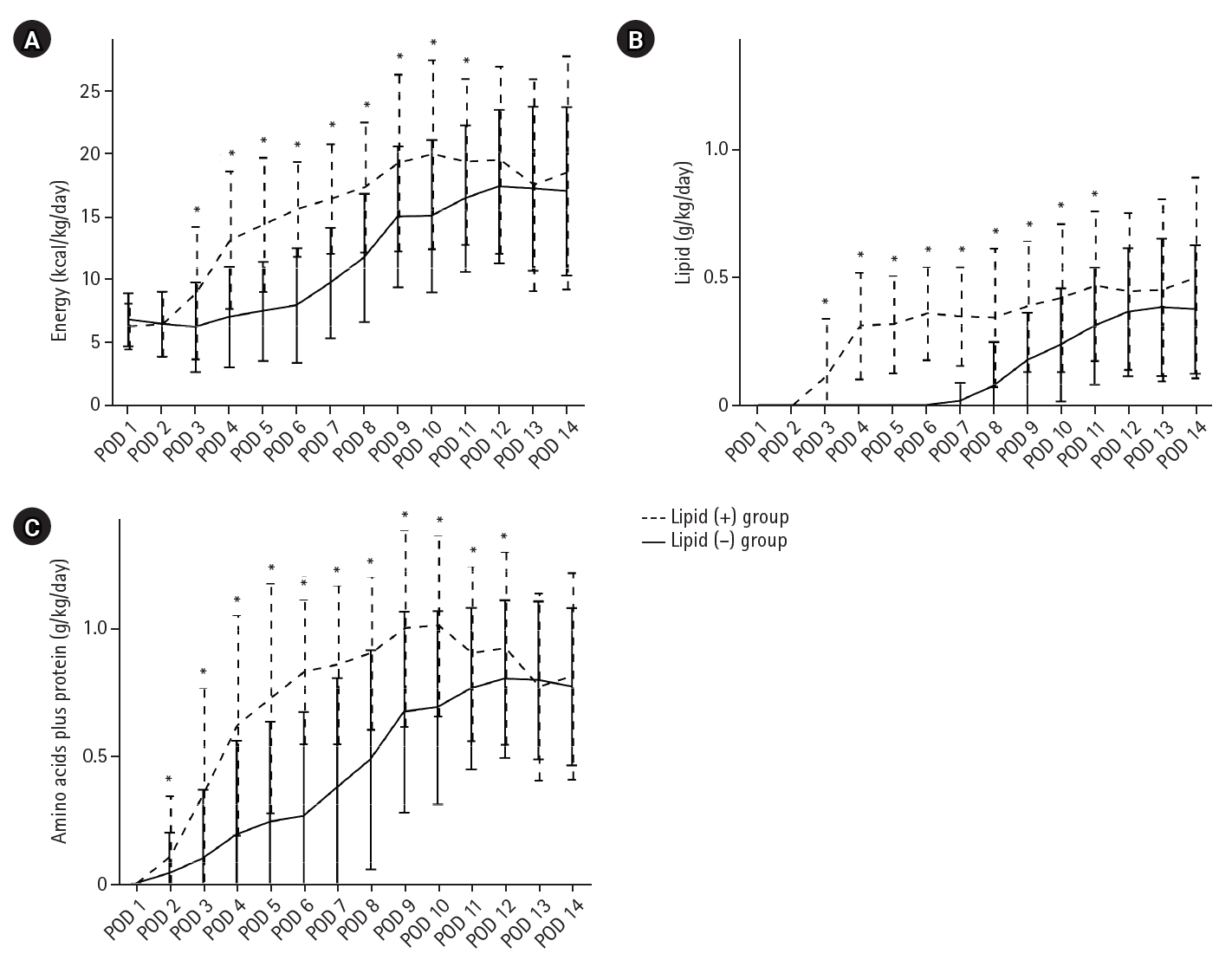
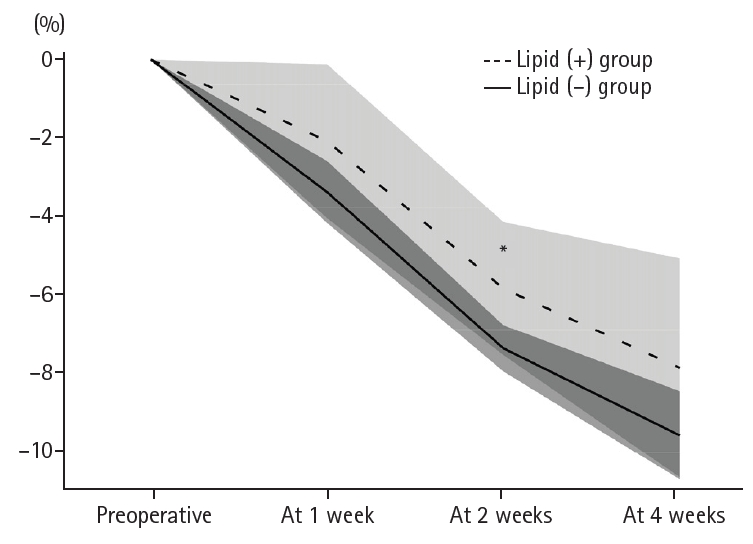
Results Patient backgrounds, surgical outcomes, and postoperative complication rates were similar between the two groups. However, patients in the lipid (+) group received significantly greater total energy and nutrient intake. PBWL at 2 weeks after surgery was significantly lower in the lipid (+) group than in the lipid (–) group (5.8% vs. 7.4%; P=0.027). Univariate analysis showed that absence of SO-ILE administration was the only significant risk factor for PBWL greater than 5% at 2 weeks after RA-TME (P=0.038).
-
Conclusion SO-ILE may have the potential to mitigate early PBWL after RA-TME.
Graphical abstract
Introduction
Methods
Surgical methods
Postoperative nutrition protocol
Results
Discussion
Authors’ contribution
Conceptualization: TN, KF, SM, RI, KK, MN, NM. Data curation: TN. Formal analysis: TN. Investigation: TN. Methodology: TN. Project administration: TN. Resources: TN. Software: TN. Supervision: KF. Validation: TN, KF, IR, KK. Visualization: TN. Writing–original draft: TN. Writing–review & editing: TN, KF, SM, RI, KK, MN, NM, SY, KY, YB. All authors read and approved the final manuscript.
Conflict of interest
The authors have no conflicts of interest regarding the publication of this article.
Funding
None.
Data availability
The raw data supporting the conclusions presented in this article will be made available by the authors upon request.
Acknowledgments
None.
Supplementary materials
Supplement 1.
Values are presented as number (%) or median (interquartile range). SPPB scores and number of sarcopenia include missing values.
BMI, body mass index; SPPB, Short Physical Performance Battery; SMI, skeletal muscle index; ASA-PS, American Society of Anesthesiologists Physical Status classification; AST, aspartate aminotransferase; ALT, alanine aminotransferase; γ-GTP, γ-glutamyl transpeptidase; CRP, C-reactive protein; NLR, neutrophil-to-lymphocyte ratio; PNI, prognostic nutritional index.
| Variable | Lipid (+) group (n=33) | Lipid (–) group (n=122) | P-value |
|---|---|---|---|
| Postoperative complicationsa | |||
| CD grade ≥II | 19 (57.6) | 69 (56.6) | 0.917 |
| CD grade ≥III | 10 (30.3) | 31 (25.4) | 0.572 |
| Pneumonia | 7 (21.2) | 18 (14.8) | 0.371 |
| Anastomotic leakage | 8 (24.2) | 19 (15.6) | 0.244 |
| Superficial surgical site infection | 3 (9.1) | 6 (4.9) | 0.363 |
| Postoperative hospital stay (day) | 22 (16–33) | 18 (17–26) | 0.774 |
| Pathological T factor | 0.672 | ||
| pT1 or 2 | 22 (66.7) | 86 (70.5) | |
| pT3 or 4 | 11 (33.3) | 36 (29.5) | |
| Lymph node metastasis | 20 (60.6) | 56 (45.9) | 0.134 |
| Presence | 13 (39.4) | 66 (54.1) | |
| Absence | |||
| Pathological stage | 0.453 | ||
| pStage I | 9 (27.3) | 47 (38.5) | |
| pStage II | 5 (15.2) | 24 (19.7) | |
| pStage III | 15 (45.5) | 40 (32.8) | |
| pStage IV | 4 (12.1) | 11 (9.0) |
| Variable | PBWL in 2 wk RA-TME, No. (%) | P-value | |
|---|---|---|---|
| ≥5% (n=106) | <5% (n=31) | ||
| Sex | 0.093 | ||
| Male | 92 (86.8) | 23 (74.2) | |
| Female | 14 (13.2) | 8 (25.8) | |
| Age ≥75 yr | 20 (18.9) | 9 (29.0) | 0.223 |
| BMI (kg/m2) | |||
| Obesity (≥25) | 17 (16.0) | 5 (16.1) | 0.990 |
| Underweight (≤18.5) | 12 (11.3) | 6 (19.4) | 0.244 |
| Preoperative chemotherapy | 52 (49.1) | 12 (38.7) | 0.310 |
| ASA-PS | 0.059 | ||
| I or II | 100 (94.3) | 26 (83.9) | |
| III | 6 (5.7) | 5 (16.1) | |
| Lymphadenectomy | 0.327 | ||
| Three fields | 22 (20.8) | 4 (12.9) | |
| Two fields | 84 (79.2) | 27 (87.1) | |
| Reconstruction route | 0.218 | ||
| Posterior mediastinal route | 101 (95.3) | 31 (100.0) | |
| Non-posterior mediastinal route | 5 (4.7) | 0 | |
| Pneumonia | 16 (15.1) | 6 (19.4) | 0.570 |
| Anastomotic leakage | 21 (19.8) | 5 (16.1) | 0.646 |
| Superficial surgical site infection | 7 (6.6) | 2 (6.5) | 0.976 |
| Pathological T factor | 0.785 | ||
| pT1 or 2 | 30 (28.3) | 8 (25.8) | |
| pT3 or 4 | 76 (71.7) | 23 (74.2) | |
| Lymph node metastasis | 0.349 | ||
| Presence | 48 (45.3) | 17 (54.84) | |
| Absence | 58 (54.7) | 14 (45.16) | |
| Postoperative complicationsa | |||
| CD grade ≥II | 62 (58.5) | 20 (64.5) | 0.547 |
| CD grade ≥III | 29 (27.4) | 10 (32.3) | 0.595 |
| Lipid use | 19 (17.9) | 11 (35.5) | 0.038 |
| Amino acid use | 45 (42.5) | 18 (58.1) | 0.125 |
| Total energy until POD 6 ≥3,000 kcal | 34 (32.1) | 14 (45.2) | 0.179 |
PBWL, postoperative body weight loss; RA-TME, robot-assisted radical transmediastinal esophagectomy; BMI, body mass index; ASA-PS, the American Society of Anesthesiologists Physical Status classification; CD, Clavien-Dindo classification; POD, postoperative day.
aCD reflects the highest-grade complication per patient.
| Multivariate analysis | ||
|---|---|---|
| OR (95% CI) | P-value | |
| Lipid-free PN | 2.22 (0.89–5.57) | 0.088 |
| Male sex | 2.12 (0.76–5.88) | 0.150 |
| ASA ≤2 | 2.93 (0.78–10.97) | 0.110 |
- 1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71:209-49. ArticlePubMedPDF
- 2. Koterazawa Y, Oshikiri T, Takiguchi G, Urakawa N, Hasegawa H, Yamamoto M, et al. Severe weight loss after minimally invasive oesophagectomy is associated with poor survival in patients with oesophageal cancer at 5 years. BMC Gastroenterol 2020;20:407.ArticlePubMedPMCPDF
- 3. Kubo Y, Miyata H, Sugimura K, Shinno N, Asukai K, Hasegawa S, et al. Prognostic implication of postoperative weight loss after esophagectomy for esophageal squamous cell cancer. Ann Surg Oncol 2021;28:184-93. ArticlePubMedPDF
- 4. Yamamoto K, Tanaka K, Yamasaki M, Yamashita K, Makino T, Saito T, et al. Early postoperative weight loss is associated with poor prognosis in patients with esophageal cancer. Esophagus 2022;19:596-603. ArticlePubMedPDF
- 5. Biere SS, van Berge Henegouwen MI, Maas KW, Bonavina L, Rosman C, Garcia JR, et al. Minimally invasive versus open oesophagectomy for patients with oesophageal cancer: a multicentre, open-label, randomised controlled trial. Lancet 2012;379:1887-92. ArticlePubMed
- 6. Cao Y, Han D, Zhou X, Han Y, Zhang Y, Li H, et al. Effects of preoperative nutrition on postoperative outcomes in esophageal cancer: a systematic review and meta-analysis. Dis Esophagus 2022;35:doab028.ArticlePubMedPDF
- 7. Low DE, Allum W, De Manzoni G, Ferri L, Immanuel A, Kuppusamy M, et al. Guidelines for perioperative care in esophagectomy: enhanced recovery after surgery (ERAS®) society recommendations. World J Surg 2019;43:299-330. ArticlePubMedPDF
- 8. Mori K, Yamagata Y, Aikou S, Nishida M, Kiyokawa T, Yagi K, et al. Short-term outcomes of robotic radical esophagectomy for esophageal cancer by a nontransthoracic approach compared with conventional transthoracic surgery. Dis Esophagus 2016;29:429-34. ArticlePubMedPDF
- 9. Yoshimura S, Mori K, Yamagata Y, Aikou S, Yagi K, Nishida M, et al. Quality of life after robot-assisted transmediastinal radical surgery for esophageal cancer. Surg Endosc 2018;32:2249-54. ArticlePubMedPMCPDF
- 10. Tashiro T, Yamamori H, Takagi K, Hayashi N, Furukawa K, Nakajima N, et al. n-3 versus n-6 polyunsaturated fatty acids in critical illness. Nutrition 1998;14:551-3. ArticlePubMed
- 11. Simopoulos AP. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother 2002;56:365-79. ArticlePubMed
- 12. Schmitz G, Ecker J. The opposing effects of n-3 and n-6 fatty acids. Prog Lipid Res 2008;47:147-55. ArticlePubMed
- 13. Kumpf VJ. Parenteral nutrition-associated liver disease in adult and pediatric patients. Nutr Clin Pract 2006;21:279-90. ArticlePubMedPDF
- 14. Lin MT, Yeh SL, Tsou SS, Wang MY, Chen WJ. Effects of parenteral structured lipid emulsion on modulating the inflammatory response in rats undergoing a total gastrectomy. Nutrition 2009;25:115-21. ArticlePubMed
- 15. Calder PC, Jensen GL, Koletzko BV, Singer P, Wanten GJ. Lipid emulsions in parenteral nutrition of intensive care patients: current thinking and future directions. Intensive Care Med 2010;36:735-49. ArticlePubMedPMC
- 16. Mori K, Yamagata Y, Wada I, Shimizu N, Nomura S, Seto Y, et al. Robotic-assisted totally transhiatal lymphadenectomy in the middle mediastinum for esophageal cancer. J Robot Surg 2013;7:385-7. ArticlePubMedPMCPDF
- 17. Brierley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours. 8th. John Wiley & Sons; 2017.
- 18. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205-13. ArticlePubMedPMC
- 19. Meguid MM, Schimmel E, Johnson WC, Meguid V, Lowell BC, Bourinski J, et al. Reduced metabolic complications in total parenteral nutrition: pilot study using fat to replace one-third of glucose calories. JPEN J Parenter Enteral Nutr 1982;6:304-7. ArticlePubMed
- 20. Thibault R, Abbasoglu O, Ioannou E, Meija L, Ottens-Oussoren K, Pichard C, et al. ESPEN guideline on hospital nutrition. Clin Nutr 2021;40:5684-709. ArticlePubMed
- 21. Battistella FD, Widergren JT, Anderson JT, Siepler JK, Weber JC, MacColl K, et al. A prospective, randomized trial of intravenous fat emulsion administration in trauma victims requiring total parenteral nutrition. J Trauma 1997;43:52-60. ArticlePubMed
- 22. McCowen KC, Friel C, Sternberg J, Chan S, Forse RA, Burke PA, et al. Hypocaloric total parenteral nutrition: effectiveness in prevention of hyperglycemia and infectious complications--a randomized clinical trial. Crit Care Med 2000;28:3606-11. ArticlePubMed
- 23. Hynes O, Anandavadivelan P, Gossage J, Johar AM, Lagergren J, Lagergren P, et al. The impact of pre- and post-operative weight loss and body mass index on prognosis in patients with oesophageal cancer. Eur J Surg Oncol 2017;43:1559-65. ArticlePubMed
References
Figure & Data
REFERENCES
Citations

- Figure
- Related articles
-
- Peripheral vein infusions of amino acids prevent early postoperative weight loss after robot-assisted radical transmediastinal esophagectomy: a retrospective study in Japan
- Association between decreased dietary intake during hospitalization and long-term weight loss in postoperative gastric cancer patients over 75 years of age: a retrospective cohort study
- Comparison of efficacy of enteral versus parenteral nutrition in patients after esophagectomy in Malaysia: a prospective cohort study
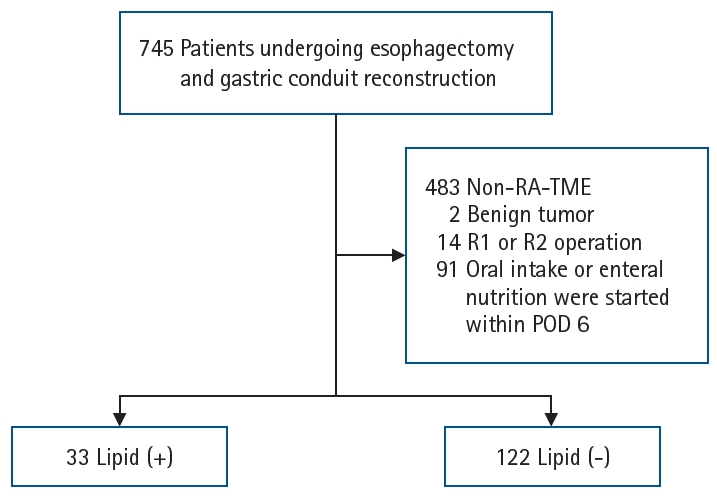
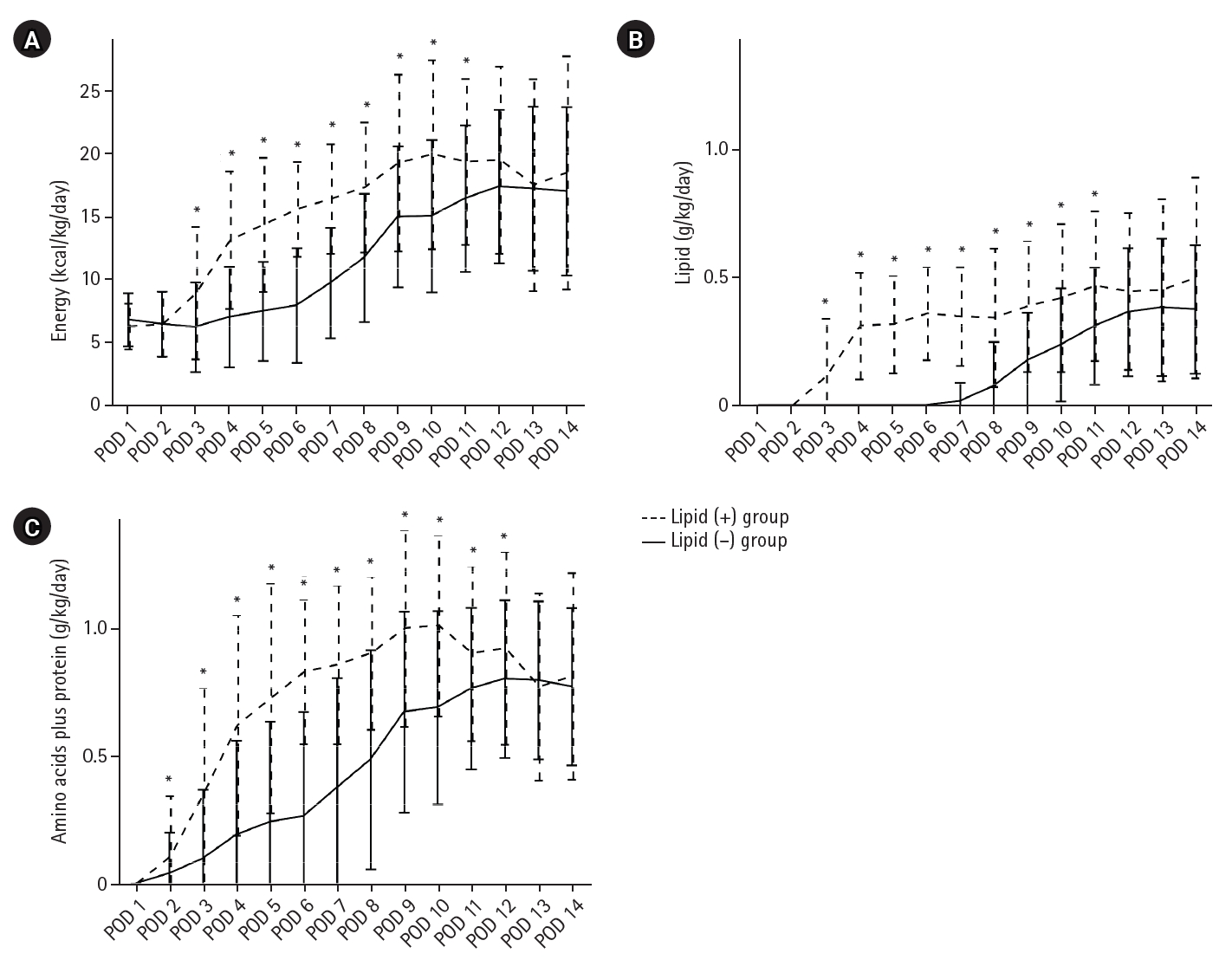
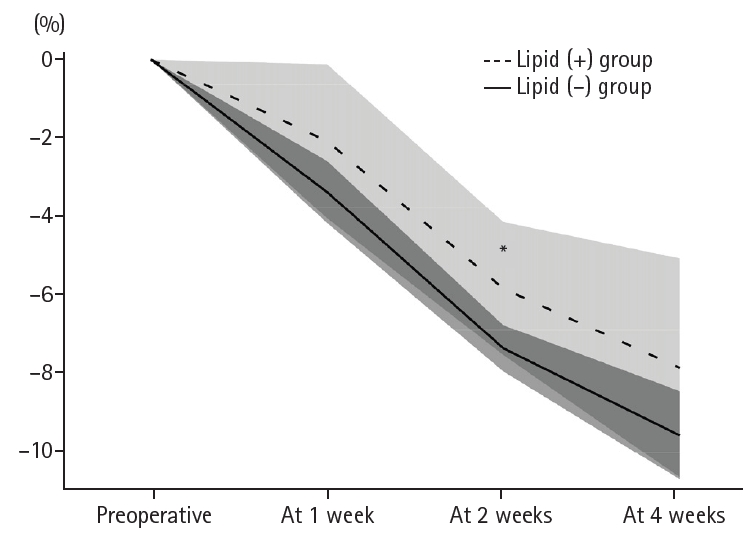
Fig. 1.
Fig. 2.
Fig. 3.
Graphical abstract
| Variable | Lipid (+) group (n=33) | Lipid (–) group (n=122) | P-value |
|---|---|---|---|
| Sex | 0.246 | ||
| Male | 26 (78.8) | 106 (86.9) | |
| Female | 7 (21.2) | 16 (13.1) | |
| Age (yr) | 68.0 (61.0–74.0) | 67.5 (60.0–73.0) | 0.627 |
| ≥75 | 7 (21.2) | 23 (18.9) | 0.761 |
| Height (cm) | 165.2 (160.1–169.9) | 166.5 (161.1–170.3) | 0.820 |
| Body weight (kg) | 59.2 (53.6–66.1) | 61.7 (53.9–67.9) | 0.266 |
| BMI (kg/m2) | 21.1 (19.4–23.8) | 22.0 (20.1–23.8) | 0.188 |
| Obesity (≥25) | 5 (15.2) | 18 (14.8) | 0.955 |
| Underweight (≤18.5) | 7 (21.2) | 14 (11.5) | 0.147 |
| Muscle mass (kg) | 45.1 (38.0–47.8) | 46.5 (42.2–49.9) | 0.118 |
| Body fat percentage (%) | 20.2 (14.7–25.6) | 19.4 (15.8–23.4) | 0.850 |
| SPPB score | 12 (12–12) | 12 (11–12) | 0.453 |
| SPPB score | 0.147 | ||
| ≤9 points | 2 (7.7) | 2 (2.0) | |
| ≥10 points | 24 (92.3) | 96 (98.0) | |
| SMI (kg/m2) | 7.3 (6.3–7.8) | 7.5 (6.8–8.1) | 0.145 |
| Low SMI | 10 (30.3) | 22 (18.0) | 0.076 |
| Sarcopenia | 6 (23.1) | 14 (13.7) | 0.241 |
| Preoperative chemotherapy | 13 (39.4) | 67 (54.9) | 0.113 |
| ASA-PS | 0.485 | ||
| I or II | 29 (87.9) | 112 (91.8) | |
| III | 4 (12.1) | 10 (8.2) | |
| White blood cells (/μL) | 6,500 (5,000–8,050) | 6,300 (5,000–7,725) | 0.873 |
| Neutrophils (/μL) | 4,543 (3,231–6,232) | 4,165 (3,239–5,201) | 0.528 |
| Lymphocytes (/μL) | 1,341 (1,079–1,578) | 1,477 (1,223–1,727) | 0.101 |
| Hemoglobin (g/dL) | 11.3 (10.1–13.4) | 12.4 (10.4–13.7) | 0.211 |
| Serum albumin (g/dL) | 4.0 (3.6–4.3) | 3.9 (3.7–4.1) | 0.825 |
| AST (U/L) | 21.0 (17.5–26.5) | 19.0 (16.0–22.0) | 0.048 |
| ALT (U/L) | 13.0 (11.0–17.5) | 14.0 (10.0–18.0) | 0.948 |
| γ-GTP (U/L) | 33.5 (21.3–49.3) | 26.0 (20.5–39.0) | 0.215 |
| Total bilirubin (mg/dL) | 0.5 (0.4–0.7) | 0.6 (0.4–0.7) | 0.678 |
| CRP (mg/dL) | 0.09 (0.04–0.27) | 0.07 (0.03–0.19) | 0.226 |
| NLR | 3.3 (2.5–4.9) | 2.8 (2.2–4.1) | 0.053 |
| PNI | 45.9 (42.4–48.5) | 46.9 (43.7–48.9) | 0.417 |
| Variable | Lipid (+) group (n=33) | Lipid (–) group (n=122) | P-value |
|---|---|---|---|
| Operation time (min) | 419 (365–458) | 434 (383–482) | 0.064 |
| Intraoperative blood loss (mL) | 160 (60–225) | 180 (90–370) | 0.057 |
| Lymphadenectomy | 0.194 | ||
| Three fields | 24 (72.7) | 101 (82.8) | |
| Two fields | 9 (27.3) | 21 (17.2) | |
| Reconstruction route | 0.643 | ||
| Posterior mediastinal route | 32 (97.0) | 116 (95.1) | |
| Non-posterior mediastinal route | 1 (3.0) | 6 (4.9) |
| Variable | Lipid (+) group (n=33) | Lipid (–) group (n=122) | P-value |
|---|---|---|---|
| Postoperative complications |
|||
| CD grade ≥II | 19 (57.6) | 69 (56.6) | 0.917 |
| CD grade ≥III | 10 (30.3) | 31 (25.4) | 0.572 |
| Pneumonia | 7 (21.2) | 18 (14.8) | 0.371 |
| Anastomotic leakage | 8 (24.2) | 19 (15.6) | 0.244 |
| Superficial surgical site infection | 3 (9.1) | 6 (4.9) | 0.363 |
| Postoperative hospital stay (day) | 22 (16–33) | 18 (17–26) | 0.774 |
| Pathological T factor | 0.672 | ||
| pT1 or 2 | 22 (66.7) | 86 (70.5) | |
| pT3 or 4 | 11 (33.3) | 36 (29.5) | |
| Lymph node metastasis | 20 (60.6) | 56 (45.9) | 0.134 |
| Presence | 13 (39.4) | 66 (54.1) | |
| Absence | |||
| Pathological stage | 0.453 | ||
| pStage I | 9 (27.3) | 47 (38.5) | |
| pStage II | 5 (15.2) | 24 (19.7) | |
| pStage III | 15 (45.5) | 40 (32.8) | |
| pStage IV | 4 (12.1) | 11 (9.0) |
| Group main effect (95% CI) | P-value | Timexgroup interaction, F (df) | P-value | Time-point contrasts | |||
|---|---|---|---|---|---|---|---|
| Time-point | Estimate (95% CI) | P-value | |||||
| White blood cells (/μL) | –398 (–764 to –33) | 0.033 | 1.31 (3,430) | 0.269 | |||
| Neutrophils (/μL) | –479 (–826 to –132) | 0.007 | 1.04 (3,426) | 0.375 | |||
| Lymphocytes (/μL) | 51 (–19 to 122) | 0.150 | 0.72 (3,419) | 0.540 | |||
| Hemoglobin (g/dL) | 0.1 (–0.2 to 0.3) | 0.604 | 1.56 (3,423) | 0.197 | |||
| Serum albumin (g/dL) | 0.0 (–0.0 to 0.1) | 0.248 | 1.31 (3,428) | 0.268 | |||
| AST (U/L) | –0.1 (–1.8 to 1.7) | 0.941 | 2.19 (3,425) | 0.089 | Week 1 | 1.5 (–0.6 to 3.6) | 0.159 |
| ALT (U/L) | 0.9 (–3.2 to 5.0) | 0.660 | 0.49 (3,427) | 0.686 | |||
| γ-GTP (U/L) | –14 (–26 to –3) | 0.180 | 2.35 (3,402) | 0.072 | Week 2 | –11 (–22 to 0) | 0.053 |
| Total bilirubin (mg/dL) | –0.0 (–0.1 to 0.1) | 0.862 | 0.58 (3,392) | 0.630 | |||
| CRP (mg/dL) | –0.7 (–1.1 to –0.3) | 0.002 | 2.78 (3,433) | 0.041 | Week 2 | –0.5 (–1.0 to 0.0) | 0.069 |
| NLR | –1.1 (–1.8 to –0.4) | 0.003 | 2.11 (3,425) | 0.098 | Week 1 | –0.5 (–1.2 to 0.1) | 0.086 |
| PNI | 0.6 (–0.2 to 1.3) | 0.132 | 0.33 (3,426) | 0.805 | |||
| Variable | PBWL in 2 wk RA-TME, No. (%) | P-value | |
|---|---|---|---|
| ≥5% (n=106) | <5% (n=31) | ||
| Sex | 0.093 | ||
| Male | 92 (86.8) | 23 (74.2) | |
| Female | 14 (13.2) | 8 (25.8) | |
| Age ≥75 yr | 20 (18.9) | 9 (29.0) | 0.223 |
| BMI (kg/m2) | |||
| Obesity (≥25) | 17 (16.0) | 5 (16.1) | 0.990 |
| Underweight (≤18.5) | 12 (11.3) | 6 (19.4) | 0.244 |
| Preoperative chemotherapy | 52 (49.1) | 12 (38.7) | 0.310 |
| ASA-PS | 0.059 | ||
| I or II | 100 (94.3) | 26 (83.9) | |
| III | 6 (5.7) | 5 (16.1) | |
| Lymphadenectomy | 0.327 | ||
| Three fields | 22 (20.8) | 4 (12.9) | |
| Two fields | 84 (79.2) | 27 (87.1) | |
| Reconstruction route | 0.218 | ||
| Posterior mediastinal route | 101 (95.3) | 31 (100.0) | |
| Non-posterior mediastinal route | 5 (4.7) | 0 | |
| Pneumonia | 16 (15.1) | 6 (19.4) | 0.570 |
| Anastomotic leakage | 21 (19.8) | 5 (16.1) | 0.646 |
| Superficial surgical site infection | 7 (6.6) | 2 (6.5) | 0.976 |
| Pathological T factor | 0.785 | ||
| pT1 or 2 | 30 (28.3) | 8 (25.8) | |
| pT3 or 4 | 76 (71.7) | 23 (74.2) | |
| Lymph node metastasis | 0.349 | ||
| Presence | 48 (45.3) | 17 (54.84) | |
| Absence | 58 (54.7) | 14 (45.16) | |
| Postoperative complications |
|||
| CD grade ≥II | 62 (58.5) | 20 (64.5) | 0.547 |
| CD grade ≥III | 29 (27.4) | 10 (32.3) | 0.595 |
| Lipid use | 19 (17.9) | 11 (35.5) | 0.038 |
| Amino acid use | 45 (42.5) | 18 (58.1) | 0.125 |
| Total energy until POD 6 ≥3,000 kcal | 34 (32.1) | 14 (45.2) | 0.179 |
| Multivariate analysis | ||
|---|---|---|
| OR (95% CI) | P-value | |
| Lipid-free PN | 2.22 (0.89–5.57) | 0.088 |
| Male sex | 2.12 (0.76–5.88) | 0.150 |
| ASA ≤2 | 2.93 (0.78–10.97) | 0.110 |
Values are presented as number (%) or median (interquartile range). SPPB scores and number of sarcopenia include missing values. BMI, body mass index; SPPB, Short Physical Performance Battery; SMI, skeletal muscle index; ASA-PS, American Society of Anesthesiologists Physical Status classification; AST, aspartate aminotransferase; ALT, alanine aminotransferase; γ-GTP, γ-glutamyl transpeptidase; CRP, C-reactive protein; NLR, neutrophil-to-lymphocyte ratio; PNI, prognostic nutritional index.
Values are presented as median (interquartile range) or number (%).
Values are presented as number (%) or median (interquartile range). Clavien-Dindo classification (CD) reflects the highest-grade complication per patient.
CI, confidence interval; AST, aspartate aminotransferase; ALT, alanine aminotransferase; γ-GTP, γ-glutamyl transpeptidase; CRP, C-reactive protein; NLR, neutrophil-to-lymphocyte ratio; PNI, prognostic nutritional index.
PBWL, postoperative body weight loss; RA-TME, robot-assisted radical transmediastinal esophagectomy; BMI, body mass index; ASA-PS, the American Society of Anesthesiologists Physical Status classification; CD, Clavien-Dindo classification; POD, postoperative day. CD reflects the highest-grade complication per patient.
PBWL, postoperative body weight loss; RA-TME, robot-assisted radical transmediastinal esophagectomy; OR, odds ratio; CI, confidence interval; PN, parenteral nutrition; ASA, American Society of Anesthesiologists.

 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN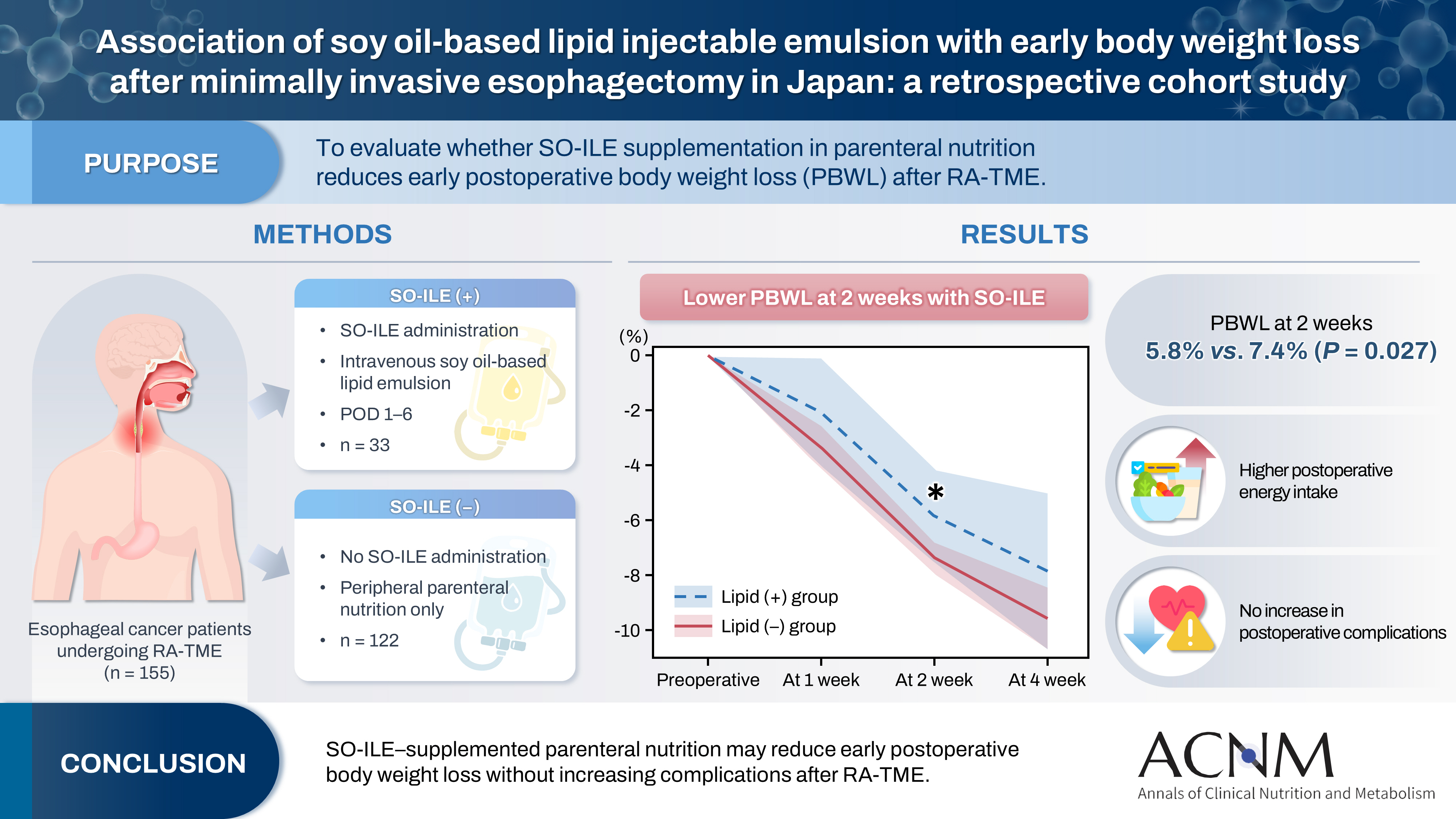
 Cite
Cite