Abstract
-
Purpose
This study focuses on the need for standardized body composition measurements in the hepatobiliary-pancreatic field. It evaluates and compares the effectiveness of bioelectrical impedance analysis (BIA) and computed tomography (CT) scans in assessing body composition of patients undergoing pancreatoduodenectomy (PD), aiming to establish correlations among different body composition indexes.
-
Methods
Ninety-seven patients who underwent PD between August 2022 and March 2023, were enrolled in this study. Muscular and fatty parameters related to BIA and CT were assessed both preoperatively and on postoperative day 6. The correlation between each parameter related to muscle fat was analyzed according to the measurement modalities.
-
Results
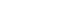
There was an increase of skeletal muscle area (SMA), total muscle area, and low attenuated muscle area after surgery. Skeletal muscle mass (SMM) measured using BIA exhibited a strong correlation with the SMA and normal attenuated muscle area (NAMA) measured using CT (r=0.86, P<0.001; r=0.76, P<0.001). The trunk muscle measured using BIA demonstrated moderate to strong correlations with SMA and NAMA measured using CT (r=0.84 P<0.001; r=0.73, P<0.001). Body fat measured using BIA and total fat area (TFA) measured using CT showed strong correlations (r=0.74, P<0.001). In the postoperative analysis, a similar trend was observed (SMM vs. SMA: r=0.80, P<0.001; SMM vs. NAMA: r=0.70, P<0.001), (trunk muscle vs. SMA: r=0.79, P<0.001; trunk muscle vs. NAMA: r=0.69, P<0.001), and (body fat vs. TFA: r=0.83, P<0.001).
-
Conclusion
BIA, akin to CT, serves as a valuable tool for assessing body composition ratios in patients undergoing PD.
-
Keywords: Body composition; Correlation of data; Electric impedance; Multidetector computed tomography; Pancreatectomy
Introduction
Background
The human body comprises different tissues, as well as water, protein, fat, and minerals. Body composition is defined as the nutritional assessment of body components [
1]. Initially, nutritionists, health professionals, and sports scientists were interested in human body composition measurements [
2].
Over the years, several studies have been conducted on how body composition is related to diseases, and many clinicians have been interested in body composition. Studies have shown that obese patients demonstrate a higher incidence of postoperative major complications [
3,
4]. Additionally, previous research has shown that sarcopenia is a negative oncological factor in patients with cancer [
5,
6]. Furthermore, body composition has been shown to exhibit strong interconnections with the short-term and long-term outcomes of several diseases.
With the rising interest in body composition, there is a corresponding need for generalization and the establishment of standardized procedures for body composition assessment. At present, various methods are available for evaluating body composition, including dual-energy X-ray absorptiometry (DEXA), computed tomography (CT), and bioelectrical impedance analysis (BIA) [
2].
Among these, a CT scan allows for accurate body composition analysis and is generally used to estimate body composition before surgery [
7,
8]. However, the cost and radiation exposure associated with CT limit their routine use for measuring body composition. Therefore, alternative methods are required to assess the body composition of patients undergoing surgery.
BIA is emerging as an alternative method for assessing body composition, with relatively accurate results reported in previous studies [
9-
11]. BIA offers several advantages over other methods, including a lower cost, a simple procedure, and the absence of radiation exposure [
12,
13].
Some cohort studies have suggested a correlation between BIA and CT visceral fat area (VFA) measurements, implying that BIA may be a potential substitute for CT body composition (CTBC) in certain patients [
14-
16]. However, other studies have shown a lack of correlation between the two methods in several disease groups. The correlation between BIA and CTBC has been limited to certain areas, indicating the need for further research in this field [
17].
Pancreatoduodenectomy (PD) is a complex surgical procedure that requires various anastomoses [
18,
19]. After surgery, patients undergo significant changes in their postoperative metabolism and body composition due to catabolism [
20]. Given these considerations, it becomes paramount to precisely assess the body composition of patients to predict surgical outcomes. However, no prior research has compared the utility of BIA and CT for evaluating body composition before and after PD.
Therefore, this study aims to assess body composition in patients who underwent PD using BIA and CTBC measurements before and after surgery and to investigate the potential areas where BIA could be a viable alternative to CTBC in hepatobiliary surgery.
Methods
Ethics statement
All the participants provided written informed consent before participating in the study. This study was approved by the Institutional Review Board (IRB) of Severance Hospital. (IRB approval number: 4-2023-0703).
Study design
Setting
Between August 2022 and March 2023, data from 97 patients who underwent PD at Severance Hospital (Seoul, Korea) were prospectively collected. BIA and CTBC were measured 6 days post-operation.
Participants
Included criteria are all patients who underwent PD at the Severance Hospital during the study period, for cure of malignancies, benign, or borderline malignancies of the stomach or duodenum. Patients who declined to participate in the study and those with contraindications to BIA, such as limb amputation or cardiac pacemakers, were excluded.
Interventions
Open or minimally invasive PD were done for surgical care of the patients.
Outcomes
Outcome variables are patients demographic findings, body composition measured by BIA and CT.
Data sources/measurement
Data are charts and laboratory and radiological findings of the patients. Each variable was measured as follows:
Clinical information collection
Patient demographics, including sex, age, height, preoperative and postoperative body weight, and body mass index (BMI), were collected through medical records and direct measurements. Preoperative laboratory tests were performed on peripheral blood samples. Detailed information regarding the surgical procedures was extracted from the surgical records documented in the medical charts.
Body composition assessment using BIA
Measurements were performed twice: on the day before surgery and postoperative day (POD) 6. The measurement protocol followed the procedures outlined in previous studies [
21]. For assessing body composition, we utilized a body water analyzer 2.0 (InBody). The skeletal muscle index (SMI) was computed by dividing the total amount of skeletal muscle mass (SMM) by the square of the individual’s height. The cut-off value of SMI for patients diagnosed with sarcopenia was <7.0 kg/m
2 in male and <5.7 kg/m
2 in female, according to the Asian Working Group for Sarcopenia: 2019 Consensus [
22].
Body composition assessment using CT
Our research institution typically conducts routine CT scans before surgery to evaluate tumor extent and resectability. To evaluate each patient’s preoperative body composition, we utilized the most recent CT scan conducted before surgery, and CT scan is conducted again on POD 6 to monitor patients’ progress after surgery. These routinely performed CT scans on POD 6 were used to analyze body composition. CT images were evaluated by an experienced gastrointestinal radiologist using the commercial software aview (version 1.1.38.6; Corelinesoft Inc.).
The components were identified on the CT slice at the L3 endplate level based on Hounsfield unit (HU) threshold values, as described in previous studies [
23]. The following ranges were designated for specific areas: −190 to 150 HU for the total muscle area (TMA), −29 to 150 HU for the skeletal muscle area (SMA), 30 to 150 HU for the normal attenuated muscle area (NAMA), −29 to 29 HU for the low attenuated muscle area (LAMA), and −190 to −30 HU for the intermuscular adipose tissue area (IMA). The relationships between muscle-related parameters measured from CT images and the areas corresponding to each parameter in the preoperative and postoperative CT images are shown in
Supplementary Fig. 1 and 2.
After generating the body mask using the pick/sculpt procedure, we identified VFA by labeling HU ranging from −190 to −30 HU, and we defined subcutaneous fat area (SFA) as HU ranging from −150 to −50 HU. The total fat area (TFA) was calculated by considering the entire area at the L3 level, including the VFA and SFA. The relationships between fat-related parameters, measured using CT images and the areas corresponding to each parameter in the preoperative and postoperative CT images, are shown in
Supplementary Fig. 2 and 3.
The SMI measured by CT was defined as the ratio of the SMA at the L3 level to the square of the patient’s height [
24]. Visceral obesity was defined as patients with VFA at L3 level ≥100 cm
2 based on the Japan Society for the Study of Obesity [
25].
Calculating the correlation of the body composition parameters according to modality
In this study, we chose three indicators linked to muscle mass assessed using BIA, including SMM, trunk muscle, and SMI. We subsequently compared these indicators with muscle mass-related parameters derived from the CT scans (TMA, NAMA, LAMA, IMA, and SMI) using Pearson’s correlation coefficient. We selected two indicators associated with fat content measured using BIA, including total body fat and VFA. Similar to the muscle-related analysis, we compared these indicators with fat-related parameters obtained from CT scans, including VFA, SFA, and TFA, using Pearson’s correlation coefficient.
Blinding (masking)
No blinding was done since there is no control group. The authors participated in the surgical intervention. They also made and analyzed a coding of the data.
Unit of analysis
All participants’ data were analyzed individually.
Bias
There was no selection bias since all target patients were included.
Statistical methods
Statistical analysis were conducted using IBM SPSS 24 (IBM Corp.) and R version 3.6.3. Continuous data were presented as the mean±standard deviation, while nominal variables were expressed as numbers and percentages. When comparing the preoperative and postoperative values of each parameter measured by CT and BIA, a paired t-test was used. The relationship between the CT and BIA parameters was evaluated using Pearson’s correlation coefficient (r). Correlation coefficients in the range of 0.90 to 1.00 were considered a “very strong correlation,” 0.70 to 0.89 as a “strong correlation,” 0.40 to 0.69 as a “moderate correlation,” and 0.10 to 0.39 as a “weak correlation” [
26].
Results
Characteristics of participants
The baseline characteristics of the participants are presented in
Table 1. A total of 97 patients who underwent PD were enrolled, including 59 males (60.8%) and 38 females (39.2%). The mean age of the patients was 62.9±11.5. The mean BMI of the patients was 23.6±3.0. Among them, 1 patient (2.0%) was classified as underweight (BMI: <18.5 kg/m
2), 67 patients (68.4%) as normal weight (BMI: 18.5 to 25.0 kg/m
2), 26 patients (26.5%) as overweight (BMI: 25.0 to 30.0 kg/m
2), and 3 patients (3.1%) as obese (BMI: ≥30.0 kg/m
2). In this study, none of the participants were diagnosed with sarcopenia, while 37 patients (38.1%) were diagnosed with visceral obesity by using CT. The median time between preoperative and subsequent CT scans on POD 6 was 8 (range, 7–15) days.
Change of body composition parameters in patients who underwent the operation
The weight and BMI of patients who underwent surgery significantly decreased after surgery (P=0.001, P=0.002, respectively). Total body fat and SMM measured using BIA significantly reduced after surgery (P=0.010, P=0.044, respectively). Similarly, TFA, VFA, and NAMA measured using CT significantly decreased after surgery (P=0.011, P=0.003, and P<0.001, respectively). In contrast, the TMA, SMA, and LAMA levels significantly increased after surgery (P=0.001, P=0.002, and P<0.001, respectively;
Table 2).
Correlation between BIA body composition and CTBC parameters
Regarding preoperative measurements, the SMM measured using BIA exhibited a strong correlation with the TMA, SMA, and NAMA measured using CT (r=0.85, P<0.001; r=0.86, P<0.001; and r=0.76, P<0.001, respectively). The trunk muscles measured using BIA exhibited moderate to strong correlations with the TMA, SMA, and NAMA measured using CT (r=0.85, P<0.001; r=0.84, P<0.001; and r=0.73, P<0.001, respectively). Additionally, body fat measured using BIA and TFA measured using CT exhibited a strong correlation (r=0.74, P<0.001), whereas VFA measured using BIA and VFA measured using CT showed a moderate correlation (r=0.53, P<0.001).
Regarding measurements after surgery, SMM measured using BIA exhibited a strong correlation with TMA, SMA, and NAMA measured using CT (r=0.77, P<0.001; r=0.80, P<0.001; and r=0.70, P<0.001, respectively). The trunk muscles measured using BIA also demonstrated strong correlations with the TMA, SMA, and NAMA measured using CT (r=0.78, P<0.001; r=0.79, P<0.001; and r=0.69, P<0.001, respectively). Body fat measured using BIA and TFA measured using CT demonstrated a strong correlation (r=0.83, P<0.001), whereas VFA measured using BIA and VFA measured using CT showed a moderate correlation (r=0.59, P<0.001;
Table 3,
Fig. 1,
Supplementary Fig. 4).
Correlation between BMI and body composition parameters based on modalities
The correlation between BMI and body composition parameters, including TFA, VFA, and body fat measured using CT or BIA was investigated. The TFA measured using CT and BMI showed a strong correlation both preoperatively and postoperatively (r=0.77, P<0.001; r=0.80, P<0.001, respectively). Body fat measured using BIA also showed a strong correlation with BMI before and after surgery (r=0.71, P<0.001; r=0.71, P<0.001, respectively). However, preoperative and postoperative VFA measured using BIA or CT exhibited a moderate correlation with BMI (r=0.58, P<0.001; r=0.65, P<0.001, respectively;
Table 4).
Discussion
Key results
In this study, we comprehensively analyzed body composition using CT and BIA measurements before and after PD. Both before and after surgery, strong correlations were observed between muscle-related indicators measured using BIA and CT. Additionally, both BIA-measured total fat and CT-measured TFA exhibited robust correlations before and after surgery, and these indicators were significantly correlated with BMI.
Interpretation/comparison with previous studies
Although the body composition measured using CT and BIA showed a reliable correlation with muscular parameters, there were some noticeable points. The first was the change in muscular parameters after surgery. Unlike the decrease in SMM measured using BIA, the TMA, SMA, and LAMA measured using CT increased after surgery. Muscle mass is commonly expected to decrease with postoperative weight loss [
20].
These divergent results could be attributed to tissue edema from surgical manipulation during abdominal surgery or overall fluid retention post-surgery [
27]. A previous study measuring the body composition of patients who underwent pancreatectomy showed an increase in total body water content, especially extracellular weight, after pancreatectomy [
28]. In our study, the mean postoperative input-output balance during the immediate postoperative period (POD 0 to 5) was approximately 4 L, which was similar to previous data.
As muscle tissue contains more water than fat tissue, postoperative tissue edema is more prominent in muscles than in fat tissue. In addition, body composition using CT measures the structured area of the patient’s image, and BIA measures impedance [
2]. Considering these two points, it was speculated that increasing muscle mass levels, such as TMA, SMA, and LAMA after surgery was not truly an increment of muscular cells, but an increase in water content caused by tissue edema, and CT could not distinguish these two aspects. This trend stood out in LAMA, which had a relatively narrow HU spectrum (−29 to 29 HU) with an increment of about 35%. Conversely, body composition using BIA could differentiate this aspect based on the underlying measurement principle.
In addition, other muscular parameters such as the NAMA score decreased after surgery in this study. Given that the HU value of water in CT scans is zero, this observation can be attributed to the NAMA (30–150 HU). Notably, this range does not include zero HU range and exhibits a muscle mass decrease that aligns with the BIA measurements, unlike SMA (−29 to 150 HU). These findings aligned with our hypotheses.
Another plausible hypothesis regarding these findings is that surgery leads to changes in muscle quality. It is well established that LAMA and IMA represent lower-quality muscles, whereas NAMA is indicative of higher-quality muscles [
23]. Following surgery, we observed a decrease in LAMA and IMA levels and an increase in NAMA levels. These findings suggest that surgery may have triggered an increase in lower-quality muscles and a decrease in higher-quality muscles, similar to the myosteatosis effect. To the best of our knowledge, no prior reports have detailed such changes in muscle quality in CT scans following surgery. However, it remains uncertain whether muscle quality can undergo significant changes within just a week after pancreatic surgery. Further studies are necessary to comprehensively explore this aspect.
Nonetheless, the SMA highly correlated with the SMM during the postoperative phase, whereas NAMA did not exhibit this correlation. These results exclusively reflected the correlation between the two parameters and not the accuracy. Further analysis, including other modalities like DEXA using different measurement strategies, is needed to establish the modality that accurately reflects the clinical status in the postoperative phase.
In the context of fat tissue before and after surgery, BIA-measured body fat demonstrated a substantial decrease post-surgery. Among the CT-based indicators, both TFA and VFA significantly decreased. These results underscore the parallel reduction in fat tissue in line with changes in body weight and BMI after surgery. Since fat tissue contains less water than muscle tissue, as previously mentioned, it was speculated that fat-related parameters were not affected by fluid therapy after surgery.
Although BIA-measured body fat exhibited a strong correlation with CT-measured TFA before and after surgery, the correlation between BIA-estimated VFA and CT-measured VFA was relatively weak. Previous studies suggested adjusted formulas using BIA-measured VFA to enhance the prediction accuracy of CT-measured VFA. This was also intended to highlight the differences in measuring principles between the two methods. Given the comparable makeup of subcutaneous and visceral fat tissues, the BIA method, which measures the body impedance, may face challenges in distinguishing between SFA and VFA. In contrast, because the discrimination between the SFA and VFA is clear in image-based measurements, measuring body composition using CT is relatively easy.
When investigating the correlations between BMI, a representative obesity index, and fat-related indicators measured using CT and BIA, CT-based TFA and BIA-measured body fat exhibited stronger correlations with BMI. The indicators measured using CT demonstrated a stronger correlation with BMI than those measured using BIA. This observation could be attributed to the capability of CT to effectively capture the overall body composition despite focusing on the fat area at the L3 level. It is plausible to interpret this finding as indicative of CT’s ability to account for the entire body’s condition, as previously reported [
7].
In summary, the body composition measured using CT and BIA demonstrated a reliable correlation. However, due to the difference in measuring principles, BIA exhibited the possibility of reliable results measuring muscle mass compared to CT in the postoperative phase. CT demonstrated a profound ability to discriminate between the VFA and SFA. This research was meaningful in proposing the first-of-its-kind selection of a suitable modality for measuring body composition based on the patient’s specific status.
Limitations
First, owing to the relative sample size, we could not perform a subgroup analysis according to sex. Occasionally, sex parameters significantly affect outcomes because of different definitions of sarcopenia based on sex. However, because the main aim of this study is to assess the correlation between modalities, this could have had little effect on the results. Second, individuals diagnosed with cancer have a different metabolism than individuals without cancer, and there can be a slight difference in the body composition between these two types of individuals [
29]. Disease-specific analysis should be performed in further studies. Also, further studies are required to identify suitable body composition measurements for specific situations.
Similar to CT, BIA is a valuable tool for assessing body composition ratios in patients who have undergone pancreatic surgery. This study evaluated the accuracy of CT and BIA for measuring body composition, which is crucial in clinical settings. Understanding the precision of these methods will pave the way for future research examining their impact on long-term survival and postoperative complications in patients treated with PD. The insights gained could lead to better management and a more accurate prediction of patient outcomes after surgery.
Supplementary materials
Supplement Fig. 1. The relathinships and CT imaging of preoperative muscle-related parameters.
Supplement Fig. 4. Preoperative and postoperative BIA-SMM vs. CT body composition parameters: scatter plots.
Acknowledgments
None.
Authors’ contribution
Conceptualization: SHK. Data curation: JK, SHK. Formal analysis: JK, SK, SHK. Funding acquisition: SHK. Investigation: SHK. Methodology: SHK, SK. Project administration: SHK. Resources: SHK, SK. Software: JK, SK, SHK. Supervision: KSK, SHK. Validation: CMK, KSK, HKH. Visualization: JK, SHK, SK. Writing – original draft: JK, SK. Writing – review & editing: all authors.
Conflict of interest
The authors of this manuscript have no conflicts of interest to disclose.
Funding
The authors received financial support from the Korean Society of Surgical Metabolism and Nutrition (No. 2021-01).
Data availability
Contact the corresponding author for data availability.
Fig. 1Scatter plots between computed tomography (CT) and bioelectrical impedance analysis (BIA) body composition parameters in preoperative and postoperative phases. (A) Preoperative skeletal muscle area (SMA) vs. skeletal muscle mass (SMM), (B) preoperative SMA vs. trunk muscle, (C) preoperative total fat vs. body fat, (D) postoperative SMA vs. SMM, (E) postoperative SMA vs. trunk muscle, and (F) postoperative total fat vs. body fat.
Table 1The baseline characteristics of participants
|
Variable |
Value (n=97) |
|
Age (yr) |
62.9±11.5 |
|
Sex |
|
|
Male |
59 (60.8) |
|
Female |
38 (39.2) |
|
Height (cm) |
164.3±9.3 |
|
Weight (kg) |
64.0±11.2 |
|
Albumin (g/dL) |
4.1±0.4 |
|
BMI (kg/m2) |
23.6±3.0 |
|
Underweight (BMI<18.5) |
1 (1.0) |
|
Normal weight (18.5≤BMI<25.0) |
67 (69.1) |
|
Overweight (25.0≤BMI<30.0) |
26 (26.8) |
|
Obese (BMI≥30.0) |
3 (3.1) |
|
Disease |
|
|
Malignancy |
77 (79.4) |
|
Benign or borderline |
20 (20.6) |
|
Operation method |
|
|
Open |
44 (45.4) |
|
Minimally invasive |
53 (54.6) |
|
Postoperative I/O (POD 0–5) (mL) |
4,848±2,090 |
|
Time interval between the CT scans (day) |
8 (7–15) |
Table 2Change of body composition parameters in patients who underwent surgery
|
Modality |
Parameter |
Preoperative |
Postoperative |
P-value |
|
Weight (kg) |
64.0±11.2 |
63.0±10.9 |
0.001 |
|
BMI (kg/m2) |
23.6±3.0 |
23.3±3.1 |
0.002 |
|
BIA |
Body fat (kg) |
16.6±6.0 |
15.8±6.3 |
0.010 |
|
SMM (kg) |
26.0±5.9 |
25.5±5.3 |
0.044 |
|
Trunk muscle (kg) |
21.2±4.7 |
20.8±3.9 |
0.123 |
|
VFA (cm2) |
76.3±34.1 |
77.5±32.6 |
0.517 |
|
CT |
TFA (cm2) |
231.3±98.8 |
222.7±95.5 |
0.011 |
|
SFA (cm2) |
140.8±59.6 |
138.3±58.5 |
0.280 |
|
VFA (cm2) |
90.5±58.4 |
84.5±56.1 |
0.003 |
|
SMA (cm2) |
126.7±29.8 |
129.9±27.7 |
0.002 |
|
TMA (cm2) |
133.1±29.6 |
137.7±29.7 |
0.001 |
|
LAMA (cm2) |
31.8±11.4 |
42.8±15.0 |
<0.001 |
|
NAMA (cm2) |
95.2±28.0 |
87.1±25.7 |
<0.001 |
|
IMA (cm2) |
6.4±4.6 |
7.8±11.8 |
0.218 |
Table 3Correlation between bioelectrical impedance analysis and computed tomography body composition parameters
|
Parameter |
|
Preoperative |
|
Postoperative |
|
BIA |
Computed tomography |
r |
P-value |
r |
P-value |
|
SMM |
Total muscle area (TMA) |
|
0.849 |
<0.001 |
|
0.765 |
<0.001 |
|
Skeletal muscle area (SMA) |
|
0.858 |
<0.001 |
|
0.800 |
<0.001 |
|
Normal attenuated muscle area (NAMA) |
|
0.763 |
<0.001 |
|
0.699 |
<0.001 |
|
Low attenuated muscle area (LAMA) |
|
0.339 |
0.001 |
|
0.278 |
0.006 |
|
Intermuscular adipose tissue area (IMA) |
|
–0.103 |
0.317 |
|
0.049 |
0.636 |
|
Trunk muscle |
Total muscle area (TMA) |
|
0.846 |
<0.001 |
|
0.778 |
<0.001 |
|
Skeletal muscle area (SMA) |
|
0.838 |
<0.001 |
|
0.793 |
<0.001 |
|
Normal attenuated muscle area (NAMA) |
|
0.728 |
<0.001 |
|
0.689 |
<0.001 |
|
Low attenuated muscle area (LAMA) |
|
0.378 |
<0.001 |
|
0.281 |
0.005 |
|
Intermuscular adipose tissue area (IMA) |
|
0.005 |
0.958 |
|
0.096 |
0.348 |
|
Body fat |
Total fat area (SFA+VFA) |
|
0.744 |
<0.001 |
|
0.826 |
<0.001 |
|
VFA |
VFA |
|
0.527 |
<0.001 |
|
0.589 |
<0.001 |
Table 4Correlation between body mass index and body composition parameters based on modalities
|
Parameter |
Preoperative |
|
Postoperative |
|
r |
P-value |
r |
P-value |
|
BMI |
CT |
Total fat |
0.77 |
<0.001 |
|
0.80 |
<0.001 |
|
|
VFA |
0.65 |
<0.001 |
|
0.64 |
<0.001 |
|
BIA |
Body fat |
0.71 |
<0.001 |
|
0.71 |
<0.001 |
|
|
VFA |
0.58 |
<0.001 |
|
0.65 |
<0.001 |
References
- 1. Willoughby D, Hewlings S, Kalman D. Body composition changes in weight loss: strategies and supplementation for maintaining lean body mass, a brief review. Nutrients 2018;10:1876.ArticlePubMedPMC
- 2. Kuriyan R. Body composition techniques. Indian J Med Res 2018;148:648-58. ArticlePubMedPMC
- 3. Tanaka K, Yamada S, Sonohara F, Takami H, Hayashi M, Kanda M, et al. Pancreatic fat and body composition measurements by computed tomography are associated with pancreatic fistula after pancreatectomy. Ann Surg Oncol 2021;28:530-8. ArticlePubMedPDF
- 4. Cullinane C, Fullard A, Croghan SM, Elliott JA, Fleming CA. Effect of obesity on perioperative outcomes following gastrointestinal surgery: meta-analysis. BJS Open 2023;7:zrad026. ArticlePubMedPMCPDF
- 5. Gortan Cappellari G, Brasacchio C, Laudisio D, Lubrano C, Pivari F, Barrea L, et al. 2022;Sarcopenic obesity: what about in the cancer setting? Nutrition 98:111624. ArticlePubMed
- 6. Kobayashi A, Kaido T, Hamaguchi Y, Okumura S, Shirai H, Yao S, et al. 2019;Impact of sarcopenic obesity on outcomes in patients undergoing hepatectomy for hepatocellular carcinoma. Ann Surg 269:924-31. ArticlePubMed
- 7. Tolonen A, Pakarinen T, Sassi A, Kyttä J, Cancino W, Rinta-Kiikka I, et al. Methodology, clinical applications, and future directions of body composition analysis using computed tomography (CT) images: a review. Eur J Radiol 2021;145:109943. ArticlePubMed
- 8. Joo I, Lee JM, Lee ES, Son JY, Lee DH, Ahn SJ, et al. 2019;Preoperative CT classification of the resectability of pancreatic cancer: interobserver agreement. Radiology 293:343-9. ArticlePubMed
- 9. Mourtzakis M, Prado CM, Lieffers JR, Reiman T, McCargar LJ, Baracos VE. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl Physiol Nutr Metab 2008;33:997-1006. ArticlePubMed
- 10. Aleixo GFP, Shachar SS, Nyrop KA, Muss HB, Battaglini CL, Williams GR. Bioelectrical impedance analysis for the assessment of sarcopenia in patients with cancer: a systematic review. Oncologist 2020;25:170-82. ArticlePubMedPDF
- 11. Xu Z, Liu Y, Yan C, Yang R, Xu L, Guo Z, et al. Measurement of visceral fat and abdominal obesity by single-frequency bioelectrical impedance and CT: a cross-sectional study. BMJ Open 2021;11:e048221. ArticlePubMedPMC
- 12. Vasold KL, Parks AC, Phelan DML, Pontifex MB, Pivarnik JM. 2019;Reliability and validity of commercially available lowcost bioelectrical impedance analysis. Int J Sport Nutr Exerc Metab 29:406-10. ArticlePubMed
- 13. Moonen HPFX, Van Zanten ARH. Bioelectric impedance analysis for body composition measurement and other potential clinical applications in critical illness. Curr Opin Crit Care 2021;27:344-53. ArticlePubMedPMC
- 14. Lee DH, Park KS, Ahn S, Ku EJ, Jung KY, Kim YJ, et al. Comparison of abdominal visceral adipose tissue area measured by computed tomography with that estimated by bioelectrical impedance analysis method in Korean subjects. Nutrients 2015;7:10513-24. ArticlePubMedPMC
- 15. Gao B, Liu Y, Ding C, Liu S, Chen X, Bian X. Comparison of visceral fat area measured by CT and bioelectrical impedance analysis in Chinese patients with gastric cancer: a cross-sectional study. BMJ Open 2020;10:e036335. ArticlePubMedPMC
- 16. Kim D, Sun JS, Lee YH, Lee JH, Hong J, Lee JM. 2019;Comparative assessment of skeletal muscle mass using computerized tomography and bioelectrical impedance analysis in critically ill patients. Clin Nutr 38:2747-55. ArticlePubMed
- 17. Hansen C, Tobberup R, Rasmussen HH, Delekta AM, Holst M. Measurement of body composition: agreement between methods of measurement by bioimpedance and computed tomography in patients with non-small cell lung cancer. Clin Nutr ESPEN 2021;44:429-36. ArticlePubMed
- 18. Aizpuru M, Starlinger P, Nagorney DM, Smoot RL, Truty MJ, Kendrick ML, et al. Contemporary outcomes of pancreaticoduodenectomy for benign and precancerous cystic lesions. HPB (Oxford) 2022;24:1416-24. ArticlePubMed
- 19. Isaji S, Mizuno S, Windsor JA, Bassi C, Fernández-Del Castillo C, Hackert T, et al. International consensus on definition and criteria of borderline resectable pancreatic ductal adenocarcinoma 2017. Pancreatology 2018;18:2-11. ArticlePubMed
- 20. Gianotti L, Besselink MG, Sandini M, Hackert T, Conlon K, Gerritsen A, et al. Nutritional support and therapy in pancreatic surgery: a position paper of the International Study Group on Pancreatic Surgery (ISGPS). Surgery 2018;164:1035-48. PubMed
- 21. Ward LC. Bioelectrical impedance analysis for body composition assessment: reflections on accuracy, clinical utility, and standardisation. Eur J Clin Nutr 2019;73:194-9. ArticlePubMedPDF
- 22. Chen LK, Woo J, Assantachai P, Auyeung TW, Chou MY, Iijima K, et al. Asian Working Group for Sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc 2020;21:300-7.e2. ArticlePubMed
- 23. Ahn H, Kim DW, Ko Y, Ha J, Shin YB, Lee J, et al. Updated systematic review and meta-analysis on diagnostic issues and the prognostic impact of myosteatosis: a new paradigm beyond sarcopenia. Ageing Res Rev 2021;70:101398. ArticlePubMed
- 24. van der Werf A, Langius JAE, de van der Schueren MAE, Nurmohamed SA, van der Pant KAMI, Blauwhoff-Buskermolen S, et al. 2018;Percentiles for skeletal muscle index, area and radiation attenuation based on computed tomography imaging in a healthy Caucasian population. Eur J Clin Nutr 72:288-96. ArticlePubMedPDF
- 25. Examination Committee of Criteria for 'Obesity Disease' in Japan; Japan Society for the Study of Obesity. New criteria for 'obesity disease' in Japan. Circ J 2002;66:987-92. ArticlePubMed
- 26. Schober P, Boer C, Schwarte LA. Correlation coefficients: appropriate use and interpretation. Anesth Analg 2018;126:1763-8. ArticlePubMed
- 27. Aoyama T. Perioperative body composition changes in the multimodal treatment of gastrointestinal cancer. Surg Today 2020;50:217-22. ArticlePubMedPDF
- 28. Petrolo M, Rangelova E, Toilou M, Hammarqvist F. Body composition, muscle function and biochemical values in patients after pancreatic surgery: an observational study. Clin Nutr 2021;40:4284-9. ArticlePubMed
- 29. Poulia KA, Sarantis P, Antoniadou D, Koustas E, Papadimitropoulou A, Papavassiliou AG, et al. Pancreatic cancer and cachexia-metabolic mechanisms and novel insights. Nutrients 2020;12:1543.ArticlePubMedPMC

 , Seung-seob Kim2
, Seung-seob Kim2 , Ho Kyoung Hwang3
, Ho Kyoung Hwang3 , Chang Moo Kang3
, Chang Moo Kang3 , Kyung Sik Kim3
, Kyung Sik Kim3 , Sung Hyun Kim3
, Sung Hyun Kim3



 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN Cite
Cite