Scopus, KCI, KoreaMed

Articles
- Page Path
- HOME > Ann Clin Nutr Metab > Volume 18(1); 2026 > Article
- Original Article Current status and short-term results regarding frailty in patients undergoing gastrointestinal cancer resection in Japan: a retrospective cohort study
-
Asuka Yasueda1
 , Junichi Nishimura2
, Junichi Nishimura2 , Seiji Ikeda3, Naotsugu Haraguchi4, Hirofumi Akita4
, Seiji Ikeda3, Naotsugu Haraguchi4, Hirofumi Akita4 , Hiroshi Wada4
, Hiroshi Wada4 , Chu Matsuda4
, Chu Matsuda4 , Takeshi Omori4
, Takeshi Omori4 , Masayoshi Yasui4
, Masayoshi Yasui4 , Hironari Tamiya5
, Hironari Tamiya5 , Hideaki Tahara6
, Hideaki Tahara6 , Hiroshi Miyata4
, Hiroshi Miyata4 -
Annals of Clinical Nutrition and Metabolism 2026;18(1):54-62.
DOI: https://doi.org/10.15747/ACNM.25.0013
Published online: March 30, 2026
1Faculty of Health and Nutrition, Otemae University, Osaka, Japan
2Department of Medical Informatics, Osaka International Cancer Institute, Osaka, Japan
3Department of Rehabilitation, Osaka International Cancer Institute, Osaka, Japan
4Department of Gastroenterological Surgery, Osaka International Cancer Institute, Osaka, Japan
5Department of Orthopaedic Surgery, Osaka International Cancer Institute, Osaka, Japan
6Department of Clinical Research Center, Osaka International Cancer Institute, Osaka, Japan
- Corresponding author: Asuka Yasueda email: yasueda@otemae.ac.jp
This article is a secondary publication of an article in Japanese published in the Japanese Journal of Surgical Metabolism and Nutrition, 2023;57(4):127-134. https://doi.org/10.11638/jssmn.57.4_127. The editors of both journals have granted permission for secondary publication.
This article includes content that was presented at the 22nd Annual Meeting of the Japanese Society of Anti-Aging Medicine and the 44th Annual Meeting of the Japanese Society of Clinical Nutrition.
© 2026 The Korean Society of Surgical Metabolism and Nutrition · The Korean Society for Parenteral and Enteral Nutrition · Asian Society of Surgical Metabolism and Nutrition
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 564 Views
- 10 Download
Abstract
-
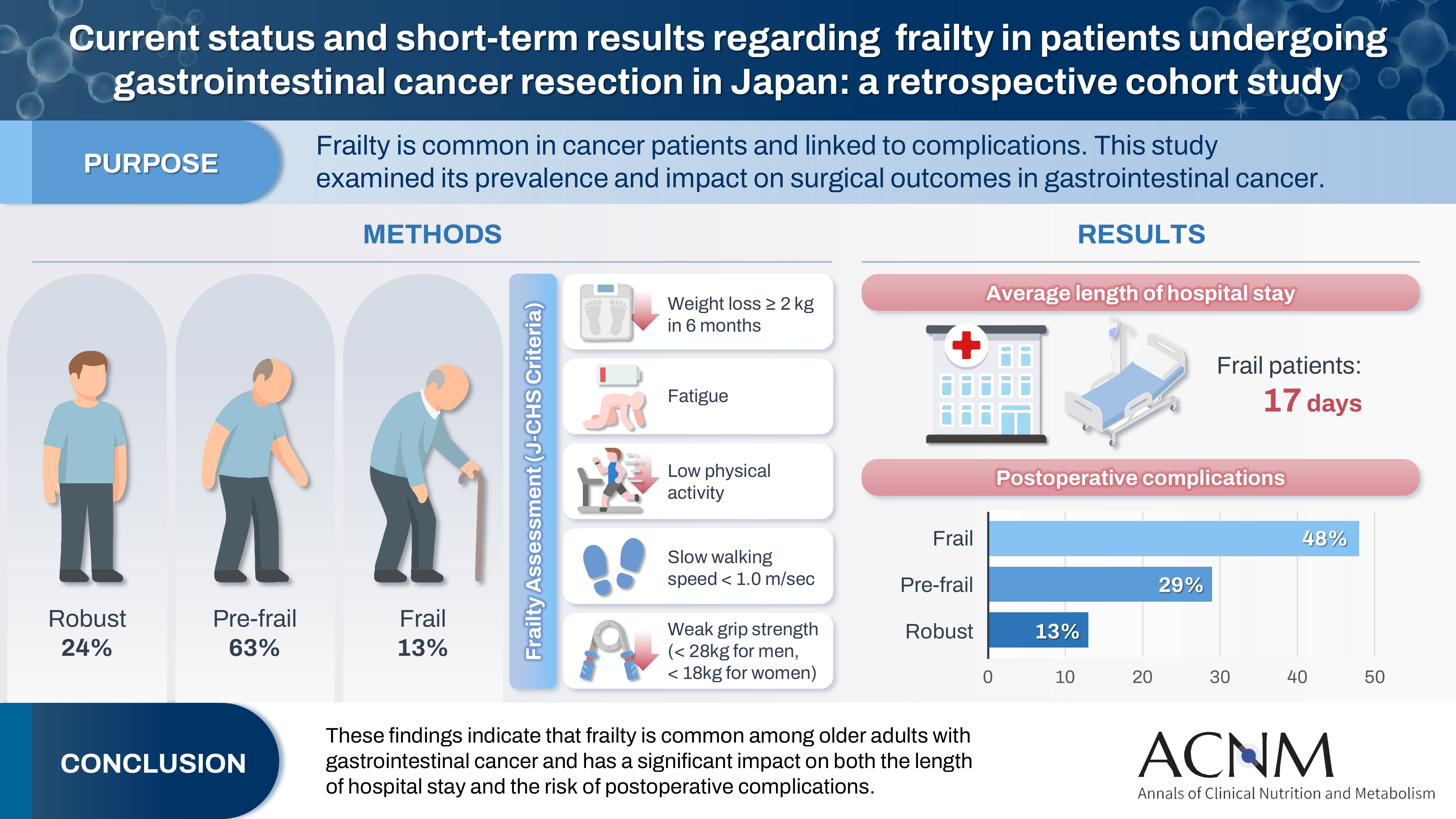
Purpose Frailty is a state of physical and cognitive decline that exists between robust health and the need for nursing care. Frailty is reported to occur at a high rate among patients with cancer and is associated with postoperative complications, such as delirium, infection, reduced survival, and rehospitalization. In this study, we investigated the incidence of frailty and surgical outcomes in patients who underwent surgery for gastrointestinal cancer.
-
Methods A total of 201 patients who underwent preoperative physical assessment at Osaka International Cancer Institute between July and September 2021 were included. The Japanese version of the Cardiovascular Health Study (J-CHS) index was used to assess the frequency of frailty and related postoperative outcomes.
-
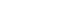
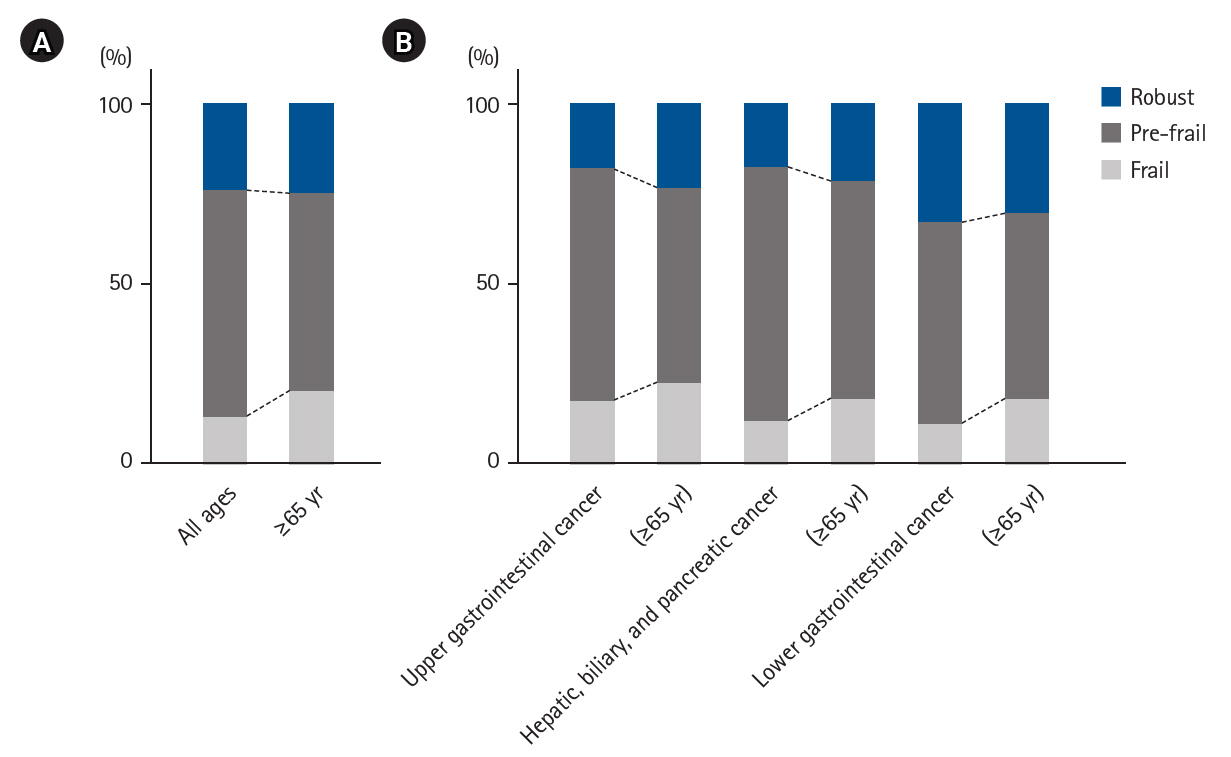
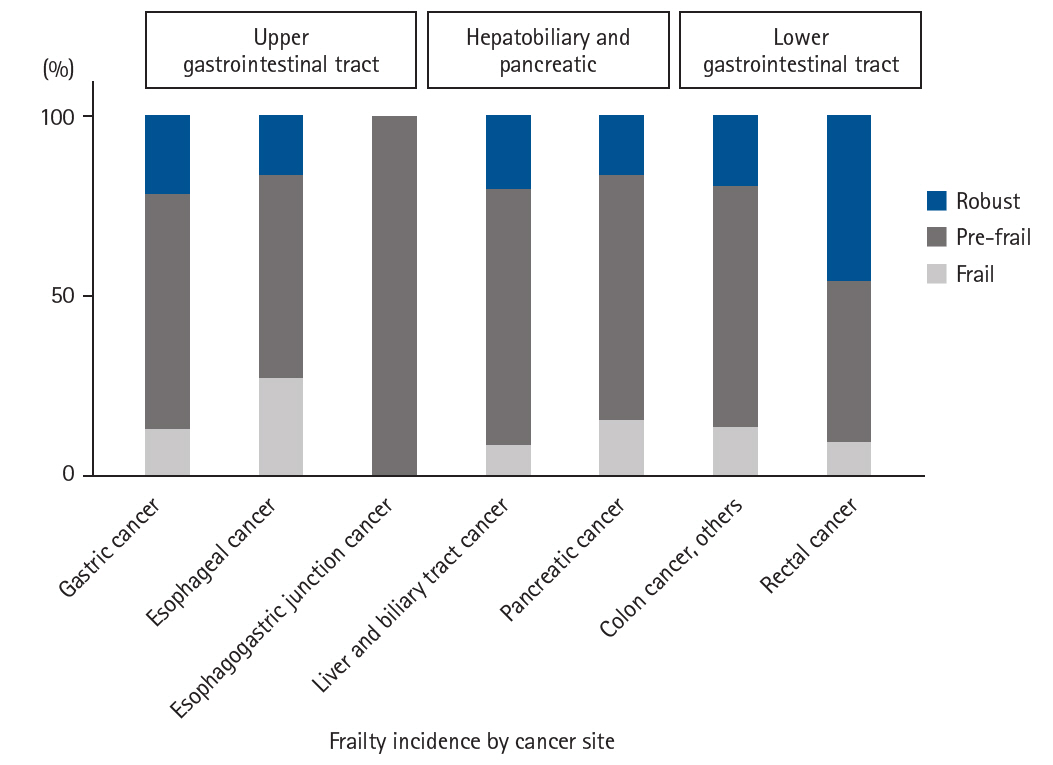
Results Among the 201 patients, 27 (13%) were classified as frail and 126 (63%) as pre-frail. Of the 27 frail patients, 22 (81%) were older adults—a significantly higher proportion compared to the pre-frail/robust group (P=0.004). The median hospital stay for frail patients was 17 days (range, 5–98 days), which was significantly longer than that for robust patients (P<0.001). Postoperative complications occurred in 15 frail patients (56%), which was higher than in pre-frail (n=40, 32%) and robust (n=6, 13%) patients. Furthermore, multivariate analysis showed that frailty was an independent risk factor for postoperative complications.
-
Conclusion These findings indicate that frailty is common among older adults with gastrointestinal cancer and has a significant impact on both the length of hospital stay and the risk of postoperative complications.
Graphical abstract
Introduction
Methods
Results
Discussion
Authors’ contribution
Conceptualization: AY, JN. Data curation: AY, SI. Formal analysis: JN, AY. Investigation: SI. Methodology: AY, CM, NH. Project administration: HA, HW. Supervision: TO, MY, HT, HT, HM. Validation: JN. Visualization: AY. Writing–original draft: AY. Writing–review: JN, NH, CM, HA, HW, TO, MY, HT, HM, HT. All authors read and approved the final manuscript.
Conflict of interest
The authors of this manuscript have no conflicts of interest to disclose.
Funding
None.
Data availability
Contact the corresponding author for data availability.
Acknowledgments
We would like to express our sincere gratitude to all the members of the Research Institute of Osaka International Cancer Center for their cooperation in conducting this study. We would also like to take this opportunity to thank everyone who supported us.
Supplementary materials
None.
- 1. Arai H. Statement from the Japan Geriatrics Society on Frailty 2014. Nihon Ronen Igakkai Zasshi 2016;53:11.
- 2. Cancer and Disease Control Division, Health Bureau, Ministry of Health, Labour and Welfare. National Cancer Registry: Incidence and Rate Report; 2019.
- 3. Hasegawa H, Takahashi A, Kakeji Y, Ueno H, Eguchi S, Endo I, et al. Surgical outcomes of gastroenterological surgery in Japan: Report of the National Clinical Database 2011-2017. Ann Gastroenterol Surg 2019;3:426-50. ArticlePubMedPMCPDF
- 4. Vacante M, Cristaldi E, Basile F, Borzi AM, Biondi A. Surgical approach and geriatric evaluation for elderly patients with colorectal cancer. Updates Surg 2019;71:411-7. ArticlePubMedPDF
- 5. Jin L, Inoue N, Sato N, Matsumoto S, Kanno H, Hashimoto Y, et al. Comparison between surgical outcomes of colorectal cancer in younger and elderly patients. World J Gastroenterol 2011;17:1642-8. ArticlePubMedPMC
- 6. Chen SY, Stem M, Cerullo M, Gearhart SL, Safar B, Fang SH, et al. The effect of frailty index on early outcomes after combined colorectal and liver resections. J Gastrointest Surg 2018;22:640-9. ArticlePubMedPDF
- 7. Indrakusuma R, Dunker MS, Peetoom JJ, Schreurs WH. Evaluation of preoperative geriatric assessment of elderly patients with colorectal carcinoma: a retrospective study. Eur J Surg Oncol 2015;41:21-7. ArticlePubMed
- 8. Satake S, Arai H. The revised Japanese version of the Cardiovascular Health Study criteria (revised J-CHS criteria). Geriatr Gerontol Int 2020;20:992-3. PubMed
- 9. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205-13. PubMedPMC
- 10. Ono R, Makiura D, Nakamura T, Okumura M, Fukuta A, Saito T, et al. Impact of preoperative social frailty on overall survival and cancer-specific survival among older patients with gastrointestinal cancer. J Am Med Dir Assoc 2021;22:1825-30. ArticlePubMed
- 11. Mima K, Miyanari N, Morito A, Yumoto S, Matsumoto T, Kosumi K, et al. Frailty is an independent risk factor for recurrence and mortality following curative resection of stage I-III colorectal cancer. Ann Gastroenterol Surg 2020;4:405-12. ArticlePubMedPMCPDF
- 12. Gietelink L, Wouters MW, Bemelman WA, Dekker JW, Tollenaar RA, Tanis PJ, et al. Reduced 30-day mortality after laparoscopic colorectal cancer surgery: a population based study from the Dutch Surgical Colorectal Audit (DSCA). Ann Surg 2016;264:135-40. PubMed
- 13. Shinall MC Jr, Arya S, Youk A, Varley P, Shah R, Massarweh NN, et al. Association of preoperative patient frailty and operative stress with postoperative mortality. JAMA Surg 2020;155:e194620.PubMedPMC
- 14. Lo BD, Leeds IL, Sundel MH, Gearhart S, Nisly GRC, Safar B, et al. Frailer patients undergoing robotic colectomies for colon cancer experience increased complication rates compared with open or laparoscopic approaches. Dis Colon Rectum 2020;63:588-97. ArticlePubMed
- 15. Murofushi K, Murakami Y, Makishima H, Moriwaki T, Sasaki T, Nakamura M, et al. Provisional clinical opinions for radiation therapy in prefrail elderly colorectal cancer patients: part two. J Jpn Soc Coloproctol 2021;74:422-29. Article
- 16. Murofushi K, Murakami Y, Makishima H, Moriwaki T, Sasaki T, Nakamura M, et al. Provisional clinical opinions for radiation therapy in prefrail elderly colorectal cancer patients: part one. J Jpn Soc Coloproctol 2021;74:413-21. Article
- 17. Mizuno I, Matsuda K, Oka S, Nishimura J, Murata K, Tanaka C, et al. Clinical recommendations for pre-frail elderly colorectal cancer patients: Clinical question—What is the appropriate anesthesia method for elderly colorectal cancer patients? Program and Abstracts of the 33rd Annual Meeting of the Japanese Society of Geriatric Anesthesiology (online meeting): 42, 2021.
- 18. Tanaka C, Mizuno I, Nishimura J, Matsuda K, Murata K, Oka S, et al. Provisional clinical opinions (PCOs) recommended by the Surgical Treatment Working Group for vulnerable elderly patients in colorectal cancer: general remarks on surgical treatment. J Jpn Soc Coloproctol 2021;74:391-400. Article
- 19. Tamura K, Karasawa K, Yamamoto H, Ogawa T, Umihori M, Watanabe K, et al. provisional clinical opinions (PCO) recommended by the Overview and Geriatric Assessment Working Group for vulnerable elderly patients in colorectal cancer: Part 2. J Jpn Soc Coloproctol 2021;74:276-86. Article
- 20. Tamura K, Karasawa K, Yamamoto H, Ogawa T, Umihori M, Watanabe K, et al. Provisional clinical opinions (PCO) Recommended by the Overview and Geriatric Assessment Working Group for vulnerable elderly patients in colorectal cancer: Part 1. J Jpn Soc Coloproctol 2021;74:269-75. Article
- 21. Ishiguro H, Ishibashi H, Yamaguchi T, Watanuki S, Hanai A, Sakurai N, et al. Provisional clinical opinions recommended by the Supportive and Palliative Care Working Group for vulnerable elderly patients with colorectal cancer. J Jpn Soc Coloproctol 2021;74:349-56. Article
- 22. Nishimura J, Yoshida Y, Tanaka C, Matsuda K, Oka S, Murata K, et al. Provisional clinical opinions (PCOs) recommended by the Surgical Treatment Working Group for vulnerable elderly patients in colorectal cancer: clinical questions with answers. J Jpn Soc Coloproctol 2021;74:401-12. Article
- 23. Fujii S, Hashimoto Y, Sakata M, Yoshimizu N, Shimamura K, Deguchi T, et al. Impact of preoperative rehabilitation interventions on perioperative outcomes in frail patients with colorectal cancer. J Jpn Soc Coloproctol 2022;75:114-23. Article
References
Figure & Data
REFERENCES
Citations

- Figure
- Related articles
-
- Association between the calcium-to-phosphorus ratio and early hypophosphatemia in preterm infants receiving parenteral nutrition in Korea: a retrospective cohort study
- The enteral feeding tube access route in esophageal cancer surgery in Japan: a retrospective cohort study
- Perioperative outcomes of older adult patients with pancreatic cancer based on nutritional status: a retrospective cohort study
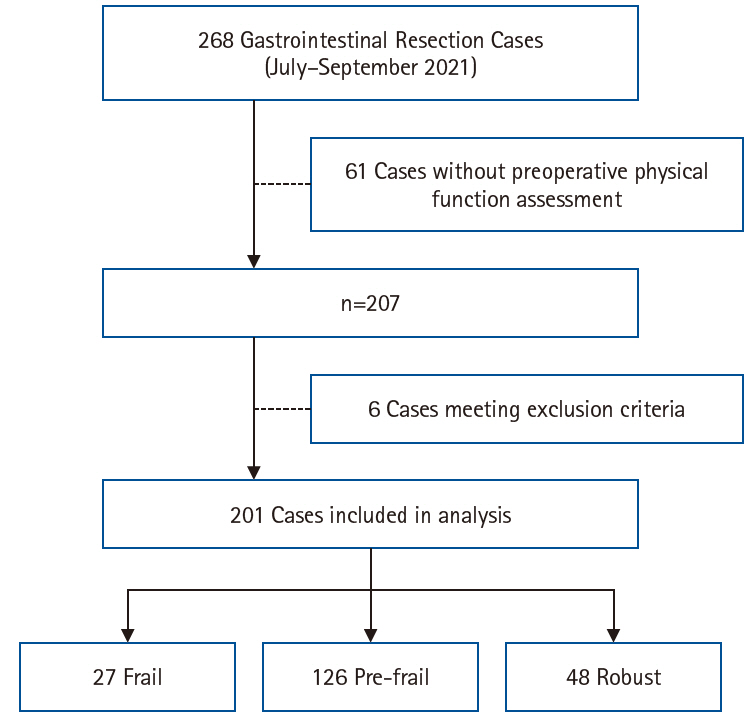
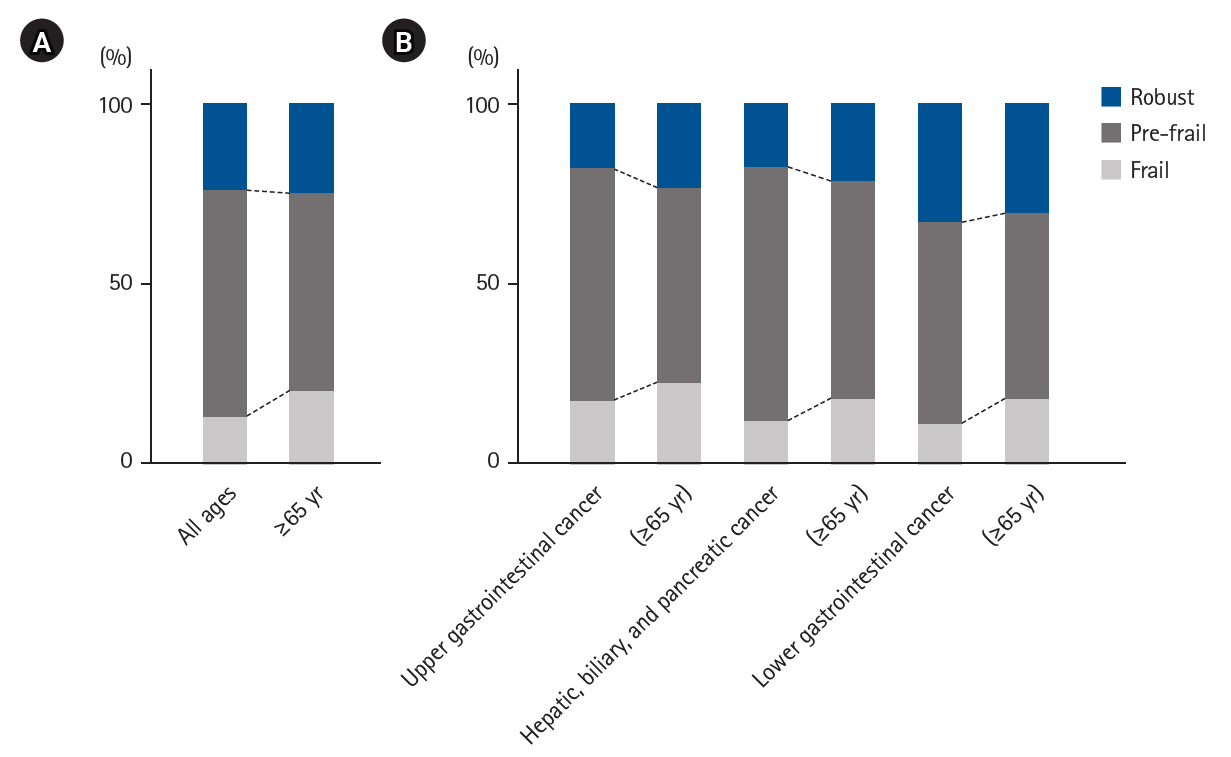
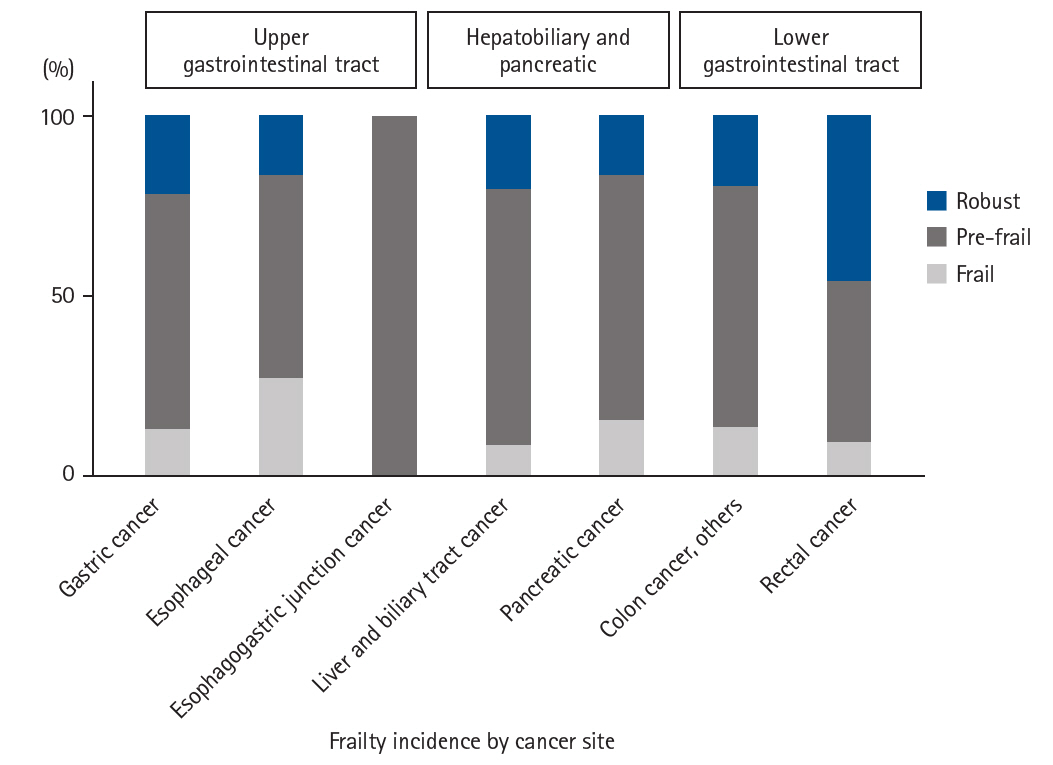
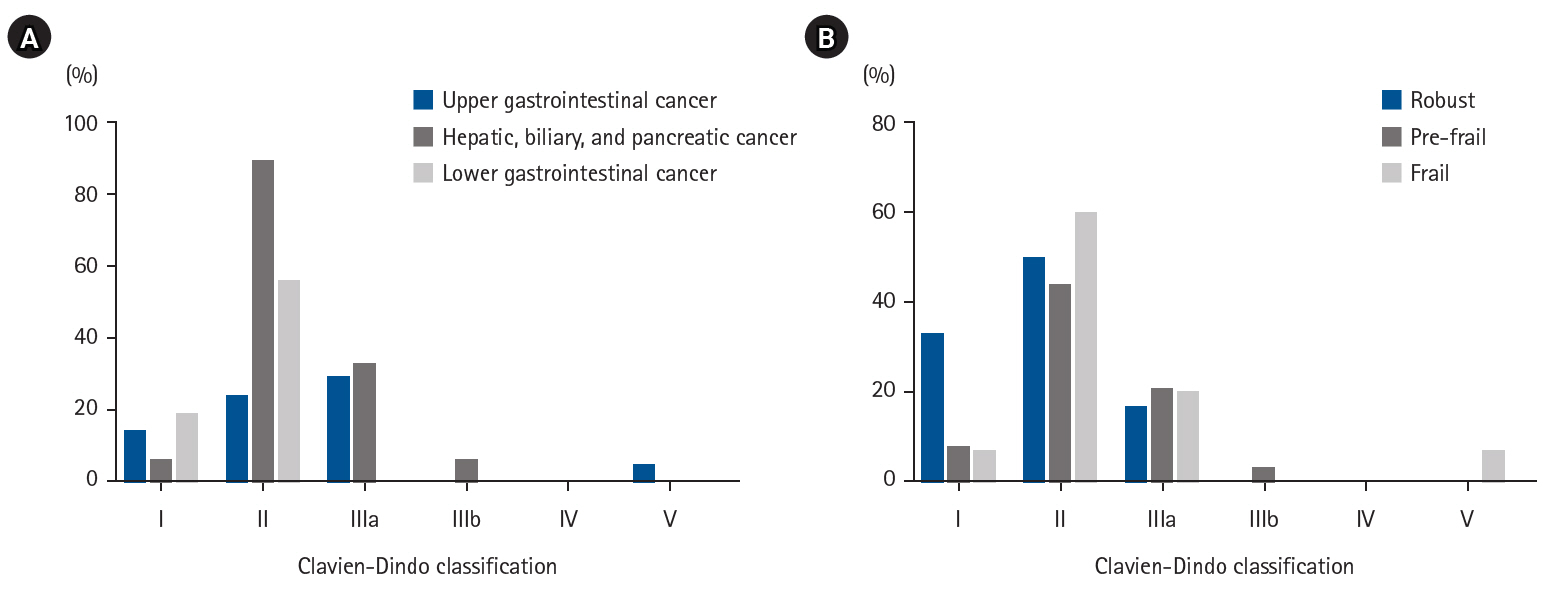
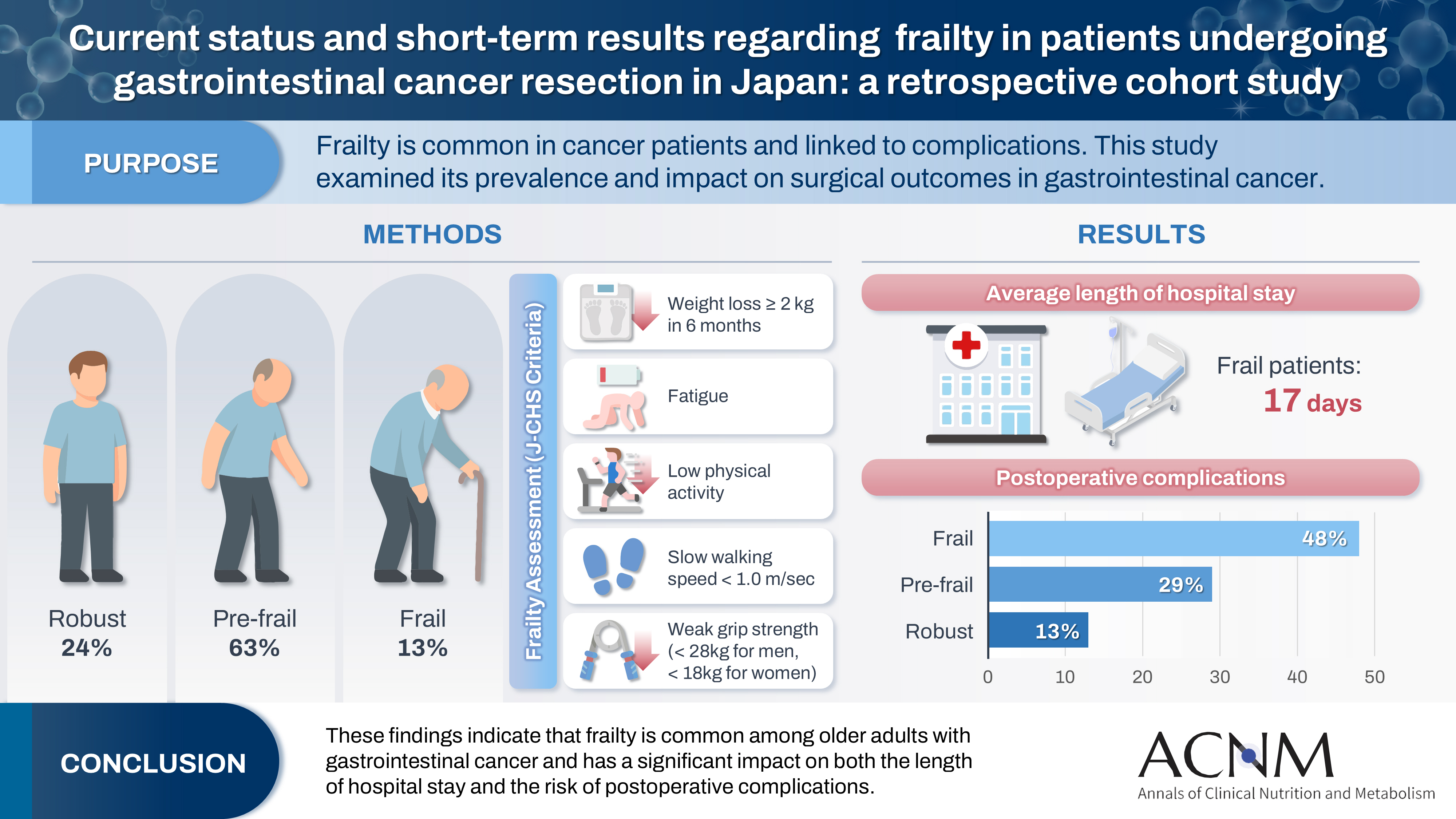
Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Graphical abstract
| Variable | Value |
|---|---|
| Sex | |
| Male | 131 (65) |
| Female | 70 (35) |
| At surgery age (yr) | 67 (28–86) |
| ≥65 yr | 112 (55) |
| Preoperative BMI (kg/m2) | 22.2 (14.1–31.6) |
| Emaciation (BMI <17 kg/m2) | 9 (4) |
| Upper GI tract | 68 (34) |
| Gastric cancer | 32 (47) |
| Esophageal cancer | 30 (44) |
| Esophagogastric junction cancer | 6 (9) |
| Hepato-biliary-pancreatic | 51 (25) |
| Liver, biliary tract cancer | 25 (49) |
| Pancreatic cancer | 26 (51) |
| Lower GI tract | 82 (41) |
| Colon cancer | 38 (46) |
| Rectal cancer | 44 (54) |
| Factor | Frail (n=27) | Pre-frail (n=126) | Robust (n=48) |
|---|---|---|---|
| Preoperative factors | |||
| Sex | |||
| Male | 13 (48) | 81 (64) | 37 (77) |
| Female | 14 (52)* | 45 (36) | 11 (33) |
| Age at surgery (≥65 yr) | 22 (81)* | 62 (49) | 28 (58) |
| Preoperative BMI (kg/m²) | 20.3 (14.1–31.1) | 22.35 (14.3–31.6) | 22.35 (18.5–31.6) |
| Emaciation (BMI <17 kg/m²) | 1 (4) | 8 (6) | 0 |
| Malnutrition (albumin <3.5 g/dL) | 6 (22)* | 11 (9) | 2 (4) |
| Intraoperative factors | |||
| Operation time (min) | 379 (167–719) | 299 (35–936) | 297 (102–687) |
| Laparoscopic surgery | 17 (63) | 74 (58) | 28 (58) |
| Robot-assisted surgery | 3 (11) | 30 (24) | 13 (27) |
| Open surgery | 7 (26) | 22 (17) | 7 (15) |
| Length of hospital stay | 17 (5–98)* | 10 (4–74) | 9 (5–52) |
| Postoperative BMI (kg/m2) | 20.3 (14.2–27.8) | 21.1 (14.5–31.9) | 21.8 (17.2–29.2) |
| Postoperative death (within 30 day) | 1 (4) | 0 | 0 |
| Postoperative complications | 15 (56)* | 40 (32) | 6 (13) |
| Cancer site/subtype | Frail | Pre-frail | Robust |
|---|---|---|---|
| Upper GI tract | |||
| Gastric cancer | 4 | 21 | 7 |
| Complications (%) | 0 | 2 (10) | 0 |
| Esophageal cancer | 8 | 17 | 5 |
| Complications (%) | 5 (63) | 9 (53) | 1 (20) |
| Esophagogastric junction cancer | 0 | 6 | 0 |
| Complications (%) | 0 | 1 (17) | 0 |
| Hepato-biliary-pancreatic | |||
| Liver, biliary tract cancer | 2 | 18 | 5 |
| Complications (%) | 1 (50) | 8 (44) | 1 (20) |
| Pancreatic cancer | 4 | 18 | 4 |
| Complications (%) | 5 (100) | 9 (50) | 1 (25) |
| Lower GI tract | |||
| Colon cancer, others | 5 | 26 | 7 |
| Complications (%) | 1 (20) | 7 (27) | 1 (14) |
| Rectal cancer | 4 | 20 | 2 |
| Complications (%) | 3 (75) | 4 (20) | 2 (10) |
| Obstructive ileus | Ureteral injury | Postoperative bleeding | Surgical site infection | Pneumonia | Recurrent laryngeal nerve palsy | Pleural effusion | Cholecystitis | Pancreatic fistula | Delayed gastric emptying | Others | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Gastric cancer | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Esophageal cancer | 0 | 0 | 0 | 3 | 4 | 4 | 1 | 0 | 0 | 0 | 4 |
| Esophagogastric junction cancer | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Liver, biliary tract cancer | 0 | 0 | 1 | 2 | 1 | 0 | 0 | 3 | 2 | 0 | 1 |
| Pancreatic cancer | 1 | 0 | 2 | 1 | 0 | 0 | 0 | 2 | 4 | 2 | 3 |
| Colon cancer, others | 2 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 3 |
| Rectal cancer | 2 | 2 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 4 |
| Univariate analysis | Multivariate analysis | |||||
|---|---|---|---|---|---|---|
| OR | 95% CI | P-value | RR | 95% CI | P-value | |
| Sex (male/female) | 1.27 | 0.663–2.518 | 0.471 | |||
| Age (≥65 yr/<65 yr) | 2.741 | 1.418–5.523 | 0.002* | 2.252 | 1.121–4.682 | 0.039* |
| Frailty (frail/pre-frail, robust) | 2.918 | 1.261–6.738 | 0.013* | 2.482 | 1.013–6.042 | 0.046* |
| Cancer site | ||||||
| Gastric cancer | 0.078 | 0.004–0.381 | <0.001 | |||
| Esophageal cancer | 1.925 | 0.889–4.080 | 0.096 | |||
| Esophagogastric junction cancer | 1.300 | 0.060–13.830 | 0.834 | |||
| Liver, biliary tract cancer | 1.037 | 0.383–2.546 | 0.939 | |||
| Pancreatic cancer | 4.602 | 1.973–11.061 | <0.001* | 3.173 | 1.294–7.983 | 0.011* |
| Colon cancer, others | 1.611 | 0.743–3.809 | 0.234 | |||
| Rectal cancer | 1.267 | 0.575–3.025 | 0.568 | |||
Values are presented as number (%) or median (range). BMI, body mass index; GI, gastrointestinal.
Values are presented as number (%) or median (range). Fisher exact test was used for comparison of numbers of cases; Dunnett’s test was used for body mass index (BMI), operation time, and hospital stay, with the robust group as control. *P<0.05
Cases with multiple complications are included.
Number of postoperative complications according to Japan Clinical Oncology Group Clavien-Dindo classification v.2.0. Cases with multiple complications are included.
OR, odds ratio; CI, confidence interval; RR, relative risk. Logistic regression was used for the analysis of risk factors for postoperative complications. *P<0.05.

 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN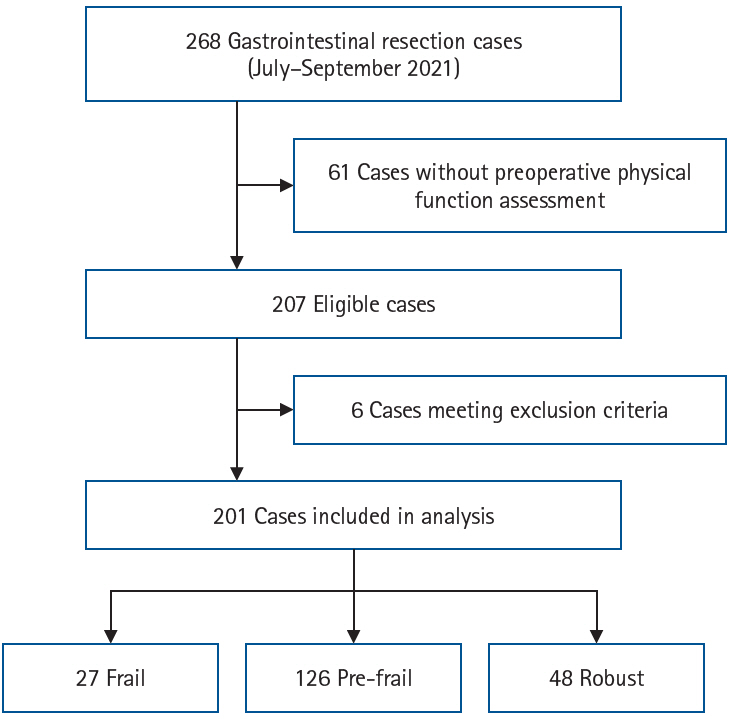
 Cite
Cite