Abstract
-
Purpose
Sarcopenia has been associated with poor outcomes in patients with cirrhosis and solid tumors. However, information is limited on the prevalence and significance of sarcopenia in patients with advanced hepatocellular carcinoma (HCC) treated with sorafenib. In the present study, we investigated the prognostic value of sarcopenia in patients with advanced HCC, treated with sorafenib after stratification by standard prognostic factors.
-
Materials and Methods
We enrolled 85 patients (77 males and 8 females) treated with sorafenib from August 2007 to August 2016. The bilateral psoas muscle area at the L3 vertebral level was measured from computed tomography images, just before sorafenib administration. This area was normalized by patients’ height, using units described in mm2/m2. Patients were classified into sarcopenia and non-sarcopenia groups using a cutoff value of 575.8 mm2/m2.
-
Results
The 3-year overall survival rate was 22.4%. Univariate analysis revealed the presence of sarcopenia (P=0.001), a baseline alpha-fetoprotein level of ≥100 ng/ml (P=0.022), derived neutrophil-to-lymphocyte ratio (P=0.019), and platelet-to-lymphocyte ratio (P=0.003) to be independent prognostic factors. In multivariate analysis, sarcopenia was independently associated with reduced overall survival (P=0.043). However, sarcopenia was not associated with reduced time on treatment.
-
Conclusion
In sorafenib treatment for advanced HCC, sarcopenia may be a significant prognostic factor and is associated with reduced survival.
-
Keywords: Hepatocelluar carcinoma; Prognostic factor; Sarcopenia
INTRODUCTION
Hepatocellular carcinoma (HCC) is one of the major causes of cancer-related mortality in the world [
1]. In Korea, HCC is the fifth most common cancer type in male and sixth in female [
2]. HCC is known to affect the elderly predominantly and is often diagnosed in advanced stage [
3,
4]. Recently, there have been remarkable developments in treatment modalities for HCC. Sorafenib, a multi-kinase inhibitor suppressing cancer growth and cell proliferation, is one of the promising treatment options, which can be used in those not amenable to transcatheter arterial chemoembolization (TACE) or liver transplantation due to HCC with an advanced stage [
5]. Sorafenib has been also recommended as first line therapy to treat HCC [
6].
There have been a number of markers which can be used to predict the prognosis after treatment with sorafenib: metastasis, vascular invasion, tumor size and other blood markers [
7]. Recently, presence of sarcopenia before sorafenib administration has been suggested as another useful marker to predict the prognosis. Due to the absence of confirmed definition to evaluate the presence of sarcopenia, however, there have been contrasting results regarding the effect of sarcopenia on the treatment results of sorafenib [
8,
9]. In addition, there is no consensus on the definition of sarcopenia in Korean population [
10], which led to the absence of studies investigating the association between sarcopenia and sorafenib in patients with HCC.
In the present study, we investigated the prognostic value of sarcopenia in patients with advanced HCC treated with sorafenib in a single tertiary center. Because of the absence of definition for sarcopenia in Korean population, we evaluated the presence of sarcopenia in the participating patients based on their median muscle mass.
MATERIALS AND METHODS
1. Patients and sorafenib therapy
This is a retrospective, single center study conducted in accordance with the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of Kyung Hee University Hospital (KHUH 2021-07-017) and informed consent was waved. 85 patients (77 males and 8 females) treated with sorafenib at Kyung Hee University Hospital between August 2007 and August 2016 were included in this study. HCC was diagnosed according to the criteria of the American Association for the Study of Liver Disease [
11] and its staging was based on abdominal dynamic contrast-enhanced computed tomography (CT) or gadolinium-enhanced magnetic resonance imaging. All patients were diagnosed as advanced HCC having Barcelona Clinic Liver Cancer (BCLC) stage C or BCLC stage B which unfit to any or failed to respond to locoregional therapies. Sorafenib was used for patients with unresectable HCC and with following features such as the presence of distant metastasis, vascular invasion, tumor thrombus in the portal vein, and refractory response to or unsuitability for TACE, transcatheter arterial infusion (TAI) chemotherapy [
12,
13]. The initial dose of sorafenib was 800 mg/day for patients with no evident risk factors, but was reduced to 400 mg/day in those with old age, low body weight, high risk of bleeding, Child-Pugh grade B, or pleural effusion/ascites [
14-
16], which was determined by the physician’s decision. Sorafenib therapy was discontinued in cases of radiologic or symptomatic progression of HCC, unacceptable sorafenib-associated toxicity or patients’ wish to discontinue the therapy. Tumor progression was evaluated according to the modified Response Evaluation Criteria in Solid Tumors [
17,
18] and/or clinically based on the presence of worsened performance status or symptoms unrelated to liver failure. In case of discontinuation, possibility of performing other therapies such as TACE, TAI or chemotherapy other than sorafenib was considered by physicians.
In order to assess the presence of sarcopenia in enrolled patients, this study utilized the CT scan data obtained 1 month prior to sorafenib administration. The skeletal muscle index (SMI) was calculated by summing cross-sectional area of bilateral psoas muscle at the level of third lumbar vertebra (measured by a hepatologist after manual tracing on the CT images) and normalizing it by the square of patients’ height [
19]. All CT images were analyzed by one observer, and the tissue Hounsfield unit (HU) limit for the skeletal muscle on CT images was between –29 HU to +150 HU [
20]. Patients were classified into either sarcopenia or non-sarcopenia group using a cutoff value of 575.8 mm
2/m
2 which is the median value of SMIs of all enrolled patients in our study (
Table 1).
Patients were classified into two groups according to the presence of sarcopenia based on the cutoff value described above and baseline characteristics of two groups were compared, expressed as mean±standard deviation or median. Student’s t-test or Mann-Whitney test was performed for group comparison. Categorical variables were described as number (percentage) and compared by using the χ2 test with Fisher’s exact test. Overall survival (OS) was calculated from the initiation date of sorafenib administration until death due to any cause or until the last follow up by using Kaplan-Meier curves and differences in the curves were assessed by the log-rank test. A Cox proportional hazards model was used for univariate analyses to determine significant factors associated with OS. The significant variables in univariate analyses were entered in multivariate analyses to find out the significant risk factors for OS in HCC patients treated with sorafenib. All statistical analyses were performed with SPSS (version 23.0; IBM Corp., Armonk, NY, USA), and a P-value<0.05 was considered statistically significant.
RESULTS
Baseline characteristics of enrolled patients are shown in
Table 1. Their median age was 60.7 (33~85) years and 77 (90.6%) of them were male. 56 patients (65.9%) had hepatitis B virus-related etiology for HCC. Baseline liver function was Child-Pugh grade A in 79 patients (92.9%) and median albumin level was 3.7±0.4 g/dL. The average duration of sorafenib administration was 3 months.
As described previously, since the enrolled patients were classified according to the median value of SMI (575.8 mm
2/m
2 [214.3~1,107.0]), the equal number of patients were included in either sarcopenic or non-sarcopenic group, and obviously SMI of two groups were significantly different (430.1±91.6 for sarcopenic group vs. 738.7±132.1 for non-sarcopenic group; P<0.001) (
Table 2). Compared with those in non-sarcopenic group, age, sex and etiology of HCC in sarcopenic group were not significantly different, but higher body mass index (BMI) in non-sarcopenic group was observed. Laboratory analysis revealed that albumin, alpha-fetoprotein (AFP), neutrophil-to-lymphocyte ratio (NLR), derived NLR (dNLR) and prognostic nutritional index were significantly different between two groups (
Table 2).
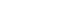
The median follow-up periods subsequent to sorafenib administration were 6.4 months for sarcopenic group and 20.6 months for non-sarcopenic group. The 3-year OS rate was 22.4%. The OS curve between two groups is shown in
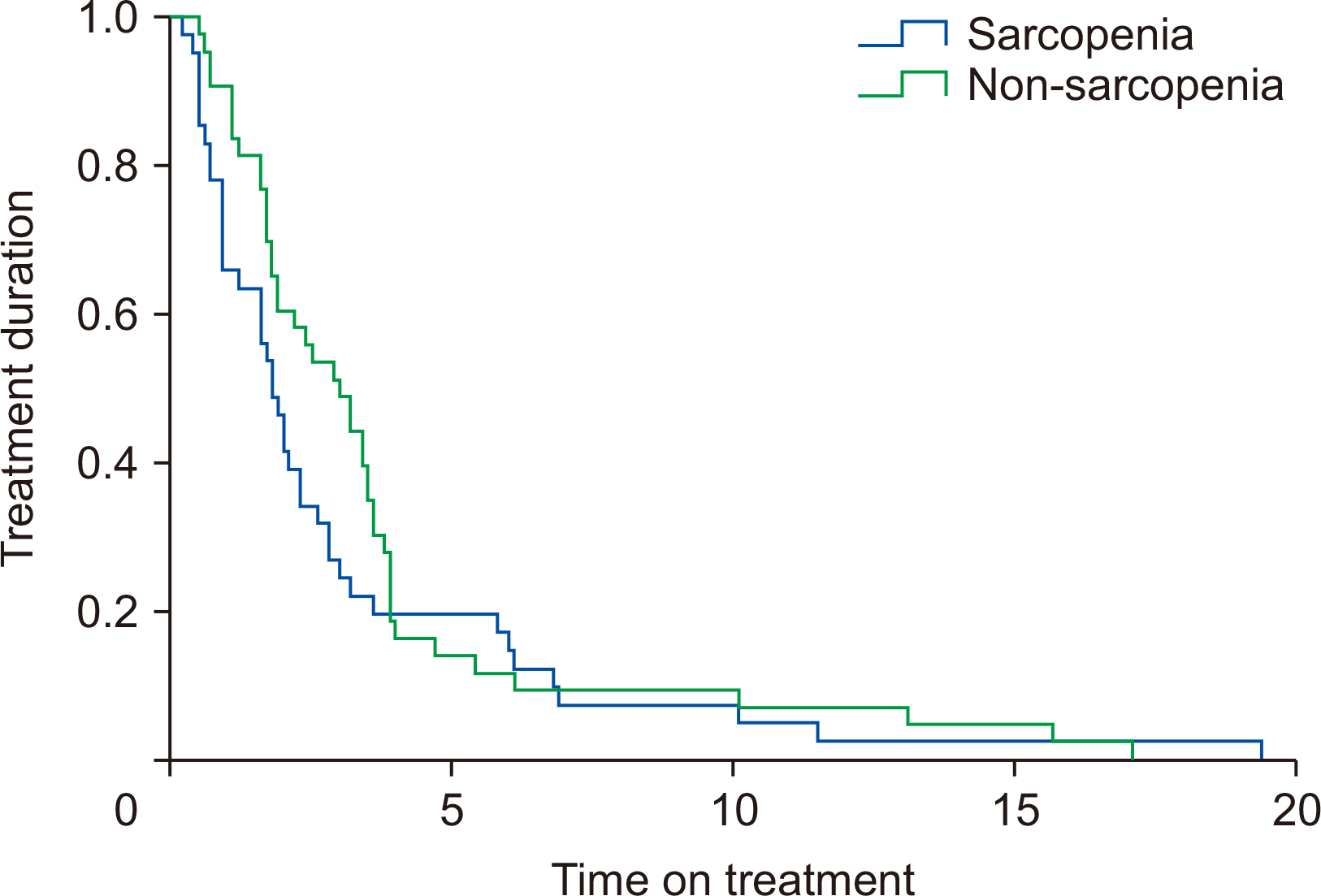
Fig. 1. The overall curve for sorafenib treatment duration between two groups is shown in
Fig. 2. According to the univariate analysis, the independent prognostic factors associated with OS were the presence of sarcopenia (P=0.001), a baseline AFP level of ≥100 ng/mL (P=0.022), dNLR (P=0.019), and PLR (P=0.003) (
Table 3). Multivariate analysis revealed that sarcopenia was the only factor independently associated with reduced OS (P=0.043), however, it was not associated with reduced time on treatment (P=0.246).
DISCUSSION
The present study was designed to investigate the usefulness of sarcopenia as a prognostic factor in patients with advanced HCC treated with sorafenib after stratification by standard prognostic factors. The 3-year OS rate was 22.4%, and it was found out that the presence of sarcopenia, a baseline AFP level (≥100 ng/mL), dNLR, and PNR were significant prognostic factors for patients with HCC enrolled in this study. Sarcopenia remained to be significant after the multivariate analysis, leading to the conclusion that sarcopenia has an independent association with reduced OS in patients with HCC receiving sorafenib therapy. However, sarcopenia was not associated with reduced time on treatment.
Previous studies have reported that the reduction of muscle mass in patients with HCC has a significant effect on the prognosis of these patients [
21,
22]. Also, the prognosis was reported to be different according to the baseline muscle mass in patients with HCC when treated with sorafenib [
8]. Therefore, it is imperative to understand the useful factors to predict the treatment effect when using sorafenib in HCC.
As described previously, there have been diverse results regarding the effect of sarcopenia in treating HCC with sorafenib. Besides the studies which demonstrated the usefulness of sarcopenia to predict the prognosis of patients treated with sorafenib [
8,
23], other study reported that the predictive power of sorafenib was only limited in those showing less than two negative prognostic factors [
7]. In addition, it was reported that sarcopenia only in those showing the reduction of mass during the treatment played a significant role as a predictive marker for future prognosis [
24].
These inconsistent data seem to be possibly due to the difference of the methods to evaluate the sarcopenia. Yamashita et al. [
24] directly measured the psoas muscle thickness to evaluate sarcopenia, while other studies used a software to measure the whole muscle mass at the L3 vertebra level [
7,
9]. Hiraoka et al. [
8] measured the whole muscle mass at the L3 vertebra level by manual calculation without using a software, which was applied in our study and two studies produced similar results. Besides the difference in measuring methods, the definition of sarcopenia varied according to the race, nation or society [
25,
26]. Our study measured the muscle mass as described by Hiraoka et al. [
8], but defined the presence of sarcopenia based on the median muscle mass of all participating patients, unlike other studies which applied certain cutoff values. Derstine et al. [
27] defined sarcopenia as 45.4 cm
2/m
2 for males and 34.4 cm
2/m
2 for females on L3 vertebra level muscle volume in American populations. In the Korean population, Sarcopenia was defined as a L3 SMI of ≤49 cm
2/m
2 for males and ≤31 cm
2/m
2 for females using Korean-specific cutoffs [
28]. However, as in the paper published by Ishii et al. [
29], the median value was used to analyze the relationship between sarcopenia and patient prognosis in this study.
For the same reason, it is not easy to find out the exact prevalence of sarcopenia in Korea, but the prevalence of sarcopenia is higher in old population in Korea [
30]. Since the prevalence of sarcopenia in cancer patients is higher than general population [
31] and the diagnosis of HCC at an old age is increasing [
2], it is expected that the number of HCC patients accompanying sarcopenia is likely to also increase, leading to the further clinical importance of sarcopenia in those treated with sorafenib.
Unlike previous studies, BMI was significantly different according to the presence or absence of sarcopenia. Also, neither extrahepatic metastasis nor vascular invasion did not demonstrate the significant effect on the prognosis. These differences may be possibly due to the different cut-off value applied for determining the presence of sarcopenia. There are some limitations in this study; our study was based on a retrospective design and did not investigate the association between the use of sorafenib and its adverse events. Median value of psoas muscle area was used as the cutoff value of sarcopenia, potentially creating bias. Also, the presence of sarcopenia was determined by manual calculation of muscle mass at the L3 vertebra level unlike previous studies [
7,
9] which used a measuring software. However, manual calculation of muscle mass was also verified by previous studies [
8,
32], and our calculation encompassed all the muscles shown in the transverse image selected for maximizing the opportunity to collect a representative muscle mass sample, which is believed not to affect the sensitivity of analysis in our study.
CONCLUSION
Sarcopenia was identified to have an independent association with reduced OS in patients with HCC receiving sorafenib therapy in Korea. Further study with larger cohort is necessary to confirm the result and compensate the limitation of this study in the future.
AUTHOR CONTRIBUTIONS
Conceptualization: MSP. Data curation: MSP, MHL. Formal analysis: MSP, MHL. Funding acquisition: MSP. Investigation: MSP. Methodology: MSP. Project administration: MSP. Resources: MSP. Software: MSP. Supervision: MSP. Validation: MSP. Visualization: MSP. Writing – original draft: MSP, MHL. Writing – review & editing: MSP.
CONFLICTS OF INTEREST
The authors of this manuscript have no conflicts of interest to disclose.
Fig. 1Kaplan–Meier curve for overall survival comparing sarcopenic and non-sarcopenic patients.
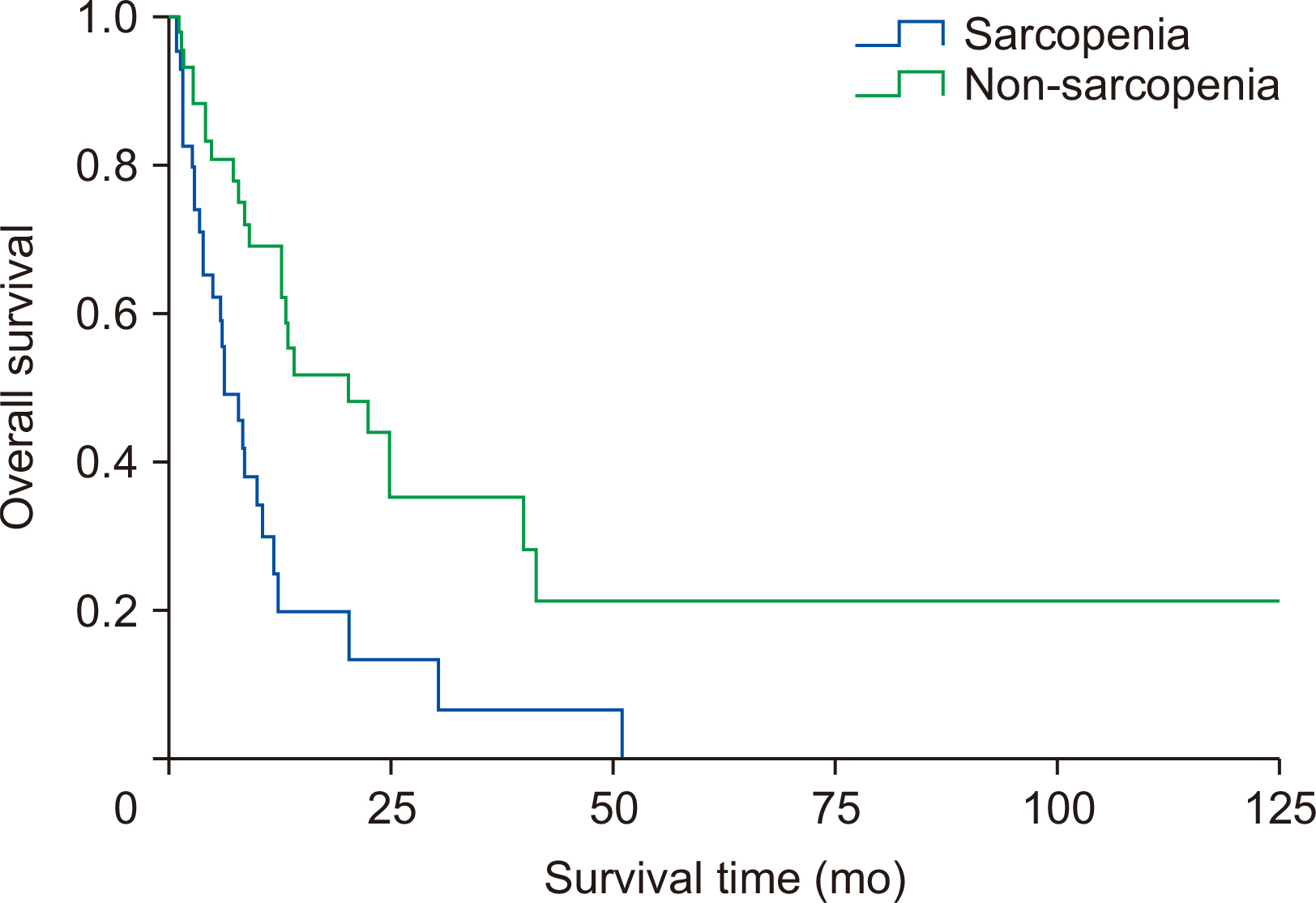
Fig. 2Kaplan–Meier curve for sorafenib treatment duration comparing sarcopenic and non-sarcopenic patients.
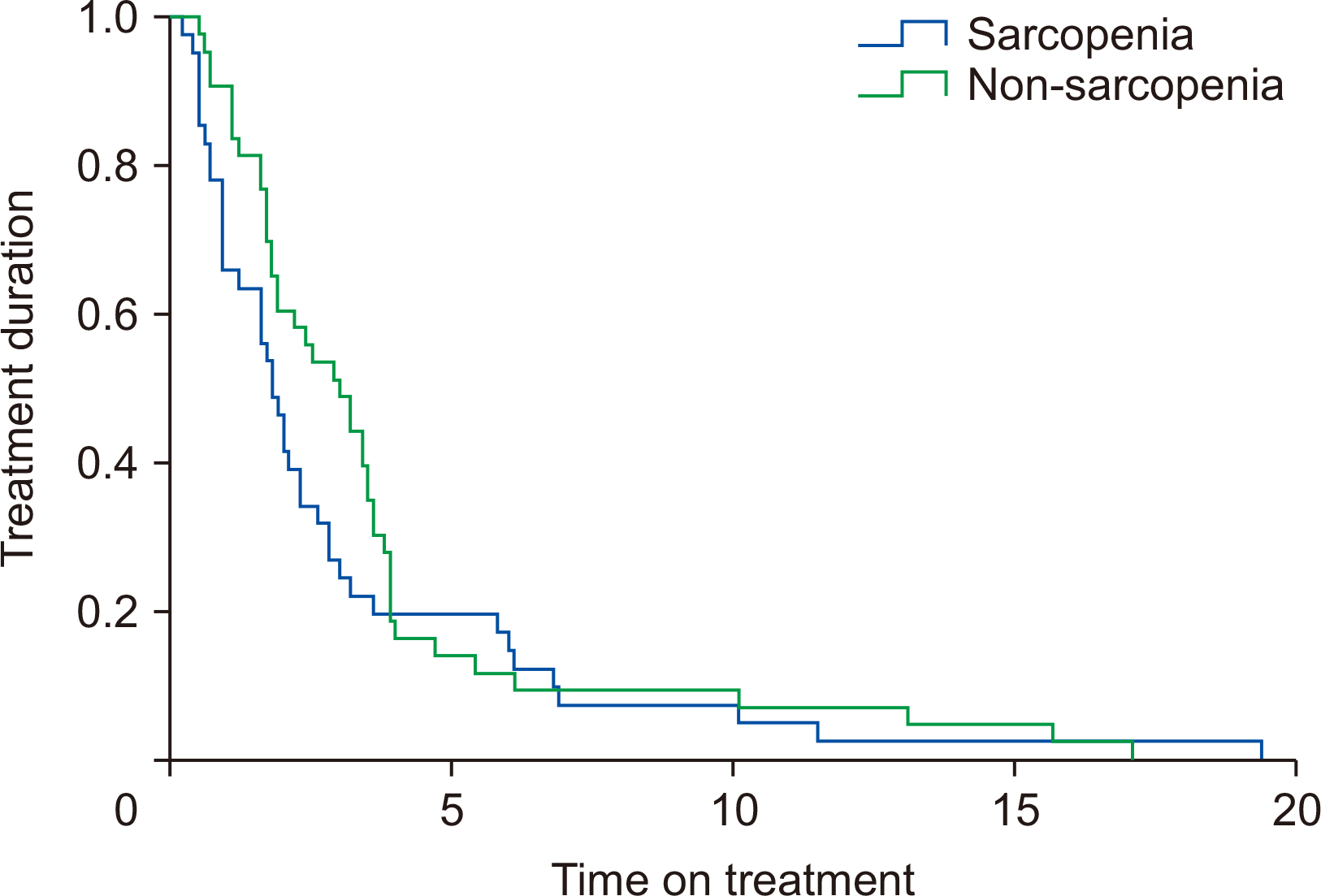
Table 1Backgrounds of patients treated with sorafenib
|
Variable |
Value |
|
Age (yr) |
60.7 (33~85) |
|
Male |
77 (90.6) |
|
Etiology |
|
|
Alcohol |
10 (11.8) |
|
HBV |
56 (65.9) |
|
HCV |
9 (10.6) |
|
Others |
10 (11.8) |
|
BMI (kg/m2) |
24.2±3.4 |
|
Albumin (g/dL) |
3.7±0.4 |
|
Total bilirubin (mg/dL) |
1.2±1.0 |
|
Prothrombin time |
1.1±0.1 |
|
AFP |
9,175.8 (1.4~60,500.0) |
|
PIVKA-II |
11,968.5 (11~75,000.0) |
|
Child-Pugh grade A |
79 (92.9) |
|
Tumor vascular invasion (+) |
40 (47.1) |
|
Extrahepatic metastasis |
49 (57.6) |
|
BCLC stage B |
29 (34.1) |
|
BCLC stage C |
56 (65.9) |
|
L3 SMI (mm2/m2) |
575.8 (214.3~1,107.0) |
Table 2Comparison of clinical and biochemical characteristics among sarcopenic and non-sarcopenic patients
|
Variable |
Sarcopenic |
Non-sarcopenic |
P-value |
|
L3 SMI |
430.1±91.6 |
738.7±132.1 |
0.001 |
|
Age (yr) |
62.6±11.6 |
59.0±10.4 |
0.143 |
|
Male |
36 (85.7) |
41 (95.3) |
0.128 |
|
Etiology |
|
|
|
|
Alcohol consumption |
6 (14.3) |
4 (9.3) |
0.853 |
|
HBV |
26 (61.9) |
30 (69.8) |
- |
|
HCV |
5 (11.9) |
4 (9.3) |
- |
|
Others |
5 (11.9) |
5 (11.6) |
- |
|
BMI (kg/m2) |
23.0±3.4 |
25.5±3.0 |
0.001 |
|
Albumin (g/dL) |
3.5±0.4 |
3.8±0.4 |
0.024 |
|
Total bilirubin (mg/dL) |
1.0±0.7 |
1.4±1.2 |
0.158 |
|
Prothrombin time |
1.1±0.1 |
1.1±0.1 |
0.423 |
|
AFP |
14,105.8 (2.4~60,500.0) |
4,435.0 (1.4~60,500.0) |
0.011 |
|
PIVKA-II |
17,193.9 (13.0~75,000.0) |
7,720.3 (11.0~75,000.0) |
0.094 |
|
Tumor vascular invasion |
21 (50.0) |
19 (44.2) |
0.591 |
|
Extrahepatic metastasis |
25 (59.5) |
24 (55.8) |
0.729 |
|
BCLC stage C |
28 (66.7) |
28 (65.1) |
0.880 |
|
NLR |
5.1±4.3 |
3.4±2.4 |
0.034 |
|
dNLR |
3.0±2.3 |
2.0±1.2 |
0.023 |
|
PLR |
197.7±145.4 |
149.6±130.8 |
0.114 |
|
ALBI |
2.0±2.1 |
2.4±1.4 |
0.408 |
|
PNI |
35.9±4.5 |
38.1±4.5 |
0.026 |
Table 3Univariate and multivariate analysis of risk factors for overall survival of hepatocellular carcinoma patients treated with sorafenib by the Cox proportional hazards model
|
Variable |
Univariate |
|
Multivariate |
|
|
|
P-value |
HR |
95% CI |
P-value |
HR |
95% CI |
|
Age (<65 yr) |
0.724 |
1.110 |
0.621~1.982 |
|
- |
- |
- |
|
Sex |
0.992 |
0.994 |
0.306~3.230 |
|
- |
- |
- |
|
BMI (<25 kg/m2) |
0.144 |
1.547 |
0.855~2.796 |
|
- |
- |
- |
|
Albumin (<3.2 g/dL) |
0.709 |
1.154 |
0.542~2.460 |
|
- |
- |
- |
|
AFP (≥100 ng/mL) |
0.022 |
1.960 |
1.096~3.507 |
|
0.105 |
1.648 |
0.901~3.015 |
|
PIVKA-II (≥40 mAU/mL) |
0.876 |
1.060 |
0.511~2.197 |
|
- |
- |
- |
|
Tumor vascular invasion |
0.448 |
1.239 |
0.709~2.164 |
|
- |
- |
- |
|
Extrahepatic metastasis |
0.173 |
0.684 |
0.394~1.188 |
|
- |
- |
- |
|
NLR (≥3.2) |
0.097 |
1.588 |
0.913~2.764 |
|
- |
- |
- |
|
dNLR (≥2.0) |
0.019 |
1.927 |
1.102~3.371 |
|
0.710 |
1.131 |
0.590~2.167 |
|
PLR (≥127.4) |
0.003 |
2.309 |
1.301~4.097 |
|
0.238 |
1.498 |
0.766~2.932 |
|
ALBI (≥2.8) |
0.296 |
0.747 |
0.430~1.297 |
|
- |
- |
- |
|
PNI (≥38) |
0.807 |
0.933 |
0.533~1.634 |
|
- |
- |
- |
|
Sarcopenia |
0.001 |
2.606 |
1.471~4.616 |
|
0.043 |
1.952 |
1.021~3.733 |
References
- 1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin 2011;61:69-90. ArticlePubMed
- 2. Kweon SS. Epidemiology of liver cancer in Korea. J Korean Med Assoc 2019;62:416-23. ArticlePDF
- 3. European Association for Study of Liver. European Organisation for Research and Treatment of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. Eur J Cancer 2012;48:599-641. ArticlePubMed
- 4. Osaki Y, Nishikawa H. Treatment for hepatocellular carcinoma in Japan over the last three decades: our experience and published work review. Hepatol Res 2015;45:59-74. PubMed
- 5. Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, et al. SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 2008;359:378-90. ArticlePubMed
- 6. Arizumi T, Ueshima K, Minami T, Kono M, Chishina H, Takita M, et al. Effectiveness of sorafenib in patients with transcatheter arterial chemoembolization (TACE) refractory and intermediate-stage hepatocellular carcinoma. Liver Cancer 2015;4:253-62. ArticlePubMedPMCPDF
- 7. Takada H, Kurosaki M, Nakanishi H, Takahashi Y, Itakura J, Tsuchiya K, et al. Impact of pre-sarcopenia in sorafenib treatment for advanced hepatocellular carcinoma. PLoS One 2018;13:e0198812. ArticlePubMedPMC
- 8. Hiraoka A, Hirooka M, Koizumi Y, Izumoto H, Ueki H, Kaneto M, et al. 2017;Muscle volume loss as a prognostic marker in hepatocellular carcinoma patients treated with sorafenib. Hepatol Res 47:558-65. ArticlePubMedPDF
- 9. Antonelli G, Gigante E, Iavarone M, Begini P, Sangiovanni A, Iannicelli E, et al. Sarcopenia is associated with reduced survival in patients with advanced hepatocellular carcinoma undergoing sorafenib treatment. United European Gastroenterol J 2018;6:1039-48. ArticlePubMedPMCPDF
- 10. Rhee SY. Role of sarcopenia in diabetes mellitus. J Korean Diabetes 2013;14:178-81. Article
- 11. Bruix J, Sherman M. American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology 2011;53:1020-2. ArticlePubMedPMC
- 12. Takeda H, Nishikawa H, Osaki Y, Tsuchiya K, Joko K, Ogawa C, et al. Japanese Red Cross Liver Study Group. Clinical features associated with radiological response to sorafenib in unresectable hepatocellular carcinoma: a large multicenter study in Japan. Liver Int 2015;35:1581-9. ArticlePubMedPDF
- 13. Nishikawa H, Takeda H, Tsuchiya K, Joko K, Ogawa C, Taniguchi H, et al. Japanese Red Cross Liver Study Group. Sorafenib therapy for BCLC stage B/C hepatocellular carcinoma; clinical outcome and safety in aged patients: a multicenter study in Japan. J Cancer 2014;5:499-509. ArticlePubMedPMC
- 14. Antoun S, Lanoy E, Iacovelli R, Albiges-Sauvin L, Loriot Y, Merad-Taoufik M, et al. Skeletal muscle density predicts prognosis in patients with metastatic renal cell carcinoma treated with targeted therapies. Cancer 2013;119:3377-84. ArticlePubMed
- 15. Morimoto M, Numata K, Kondo M, Hidaka H, Takada J, Shibuya A, et al. Higher discontinuation and lower survival rates are likely in elderly Japanese patients with advanced hepatocellular carcinoma receiving sorafenib. Hepatol Res 2011;41:296-302. ArticlePubMed
- 16. Antoun S, Baracos VE, Birdsell L, Escudier B, Sawyer MB. Low body mass index and sarcopenia associated with dose-limiting toxicity of sorafenib in patients with renal cell carcinoma. Ann Oncol 2010;21:1594-8. ArticlePubMed
- 17. Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009;45:228-47. ArticlePubMed
- 18. Lencioni R, Llovet JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis [Epub]. https://doi.org/10.1055/s-0030-1247132. Feb 19;Article
- 19. Jin WH, Mellon EA, Frakes JM, Murimwa GZ, Hodul PJ, Pimiento JM, et al. Impact of sarcopenia in borderline resectable and locally advanced pancreatic cancer patients receiving stereotactic body radiation therapy. J Gastrointest Oncol 2010;2018;9:24-34. ArticlePubMedPMC
- 20. Harimoto N, Shirabe K, Yamashita YI, Ikegami T, Yoshizumi T, Soejima Y, et al. Sarcopenia as a predictor of prognosis in patients following hepatectomy for hepatocellular carcinoma. Br J Surg 2013;100:1523-30. ArticlePubMedPDF
- 21. Montano-Loza AJ, Meza-Junco J, Prado CM, Lieffers JR, Baracos VE, Bain VG, et al. Muscle wasting is associated with mortality in patients with cirrhosis. Clin Gastroenterol Hepatol Sep 3;[Epub]. https://doi.org/10.1016/j.cgh.2011.08.028. Article
- 22. Hanai T, Shiraki M, Nishimura K, Ohnishi S, Imai K, Suetsugu A, et al. Sarcopenia impairs prognosis of patients with liver cirrhosis. Nutrition 2011;2015;31:193-9. ArticlePubMed
- 23. Nishikawa H, Nishijima N, Enomoto H, Sakamoto A, Nasu A, Komekado H, et al. Prognostic significance of sarcopenia in patients with hepatocellular carcinoma undergoing sorafenib therapy. Oncol Lett 2017;14:1637-47. ArticlePubMedPMC
- 24. Yamashima M, Miyaaki H, Honda T, Shibata H, Miuma S, Taura N, et al. Significance of psoas muscle thickness as an indicator of muscle atrophy in patients with hepatocellular carcinoma treated with sorafenib. Mol Clin Oncol 2017;7:449-53. ArticlePubMedPMC
- 25. Martin L, Birdsell L, Macdonald N, Reiman T, Clandinin MT, McCargar LJ, et al. Cancer cachexia in the age of obesity: skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J Clin Oncol 2013;31:1539-47. ArticlePubMed
- 26. Nishikawa H, Shiraki M, Hiramatsu A, Moriya K, Hino K, Nishiguchi S. Japan Society of Hepatology guidelines for sarcopenia in liver disease (1st edition): recommendation from the working group for creation of sarcopenia assessment criteria. Hepatol Res 2016;46:951-63. ArticlePubMedPDF
- 27. Derstine BA, Holcombe SA, Ross BE, Wang NC, Su GL, Wang SC. Skeletal muscle cutoff values for sarcopenia diagnosis using T10 to L5 measurements in a healthy US population. Sci Rep 2018;8:11369.ArticlePubMedPMCPDF
- 28. Lee JS, Kim YS, Kim EY, Jin W. Prognostic significance of CT-determined sarcopenia in patients with advanced gastric cancer. PLoS One 2018;13:e0202700. ArticlePubMedPMC
- 29. Ishii N, Iwata Y, Nishikawa H, Enomoto H, Aizawa N, Ishii A, et al. 2017;Effect of pretreatment psoas muscle mass on survival for patients with unresectable pancreatic cancer undergoing systemic chemotherapy. Oncol Lett 14:6059-65. ArticlePubMedPMC
- 30. Kim TN, Yang SJ, Yoo HJ, Lim KI, Kang HJ, Song W, et al. Prevalence of sarcopenia and sarcopenic obesity in Korean adults: the Korean sarcopenic obesity study. Int J Obes (Lond) 2009;33:885-92. ArticlePubMedPDF
- 31. Moon JH, Kong MH, Kim HJ. Prevalence of sarcopenia and its association with metabolic syndrome in Korean cancer survivors. Korean J Obes 2015;24:140-7. Article
- 32. Hiraoka A, Aibiki T, Okudaira T, Toshimori A, Kawamura T, Nakahara H, et al. Muscle atrophy as pre-sarcopenia in Japanese patients with chronic liver disease: computed tomography is useful for evaluation. J Gastroenterol 2015;50:1206-13. ArticlePubMedPMCPDF

 , Min-Su Park, M.D., Ph.D.
, Min-Su Park, M.D., Ph.D.


 E-submission
E-submission KSPEN
KSPEN KSSMN
KSSMN ASSMN
ASSMN JSSMN
JSSMN

 Cite
Cite